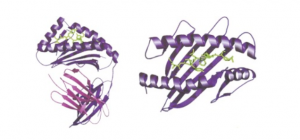
As one of the two primary classes of major histocompatibility complex (MHC) molecules, MHC class I molecules are expressed in almost all cell types and can present peptide antigens to CD8+ T lymphocytes, which helps to trigger an immediate response from the immune system. In terms of structure, MHC class I molecules consist of a single α-chain and associate with β2-microglobulin for proper folding and trafficking to the cell surface. The following figure shows how an MHC class I molecule bind a virally derived peptide.
Although MHC class I molecules contribute greatly in alerting the immune system to virally infected cells, it is still not well-understood as to how MHC-1 molecules bind to antigen peptides in the human immune system. Now this puzzle is probably clarified as recently researchers at the University of California, Santa Cruz have unveiled the details of key molecular interactions related to MHC-1 protein selection and processing of antigens. Their new findings, published in the December 3 issue of PNAS.
As is previously known, the role of the MHC-1 protein is to enable every cell in the body to display on its surface fragments of all the proteins that are being produced in that cell. With the help of MHC-1 proteins, cytotoxic T cells can more easily recognize foreign proteins derived from pathogens or mutant proteins produced by tumor tissues and make quick immune responses accordingly.
Sgourakis, assistant professor of chemistry and biochemistry at the University of California, Santa Cruz, and his team worked closely with Erik Procko’s group at the University of Illinois to understand the mechanism of protein-peptide selection and binding to MHC-I proteins. This new paper reveals how the interaction between MHC-1 protein and molecular chaperone affects the composition of displayed antigens.
There are thousands of different variants of the human MHC-1 protein, produced by different “alleles” of the MHC-1 gene. The polymorphism of MHC-1 protein is the cause of many individual differences in the immune response, including differences in sensitivity to autoimmune diseases, infections, and cancer. Each person has six major MHC-1 alleles (three from the mother and three from the father). These six MHC-I proteins can load all the peptides produced in a cell.
Sgourakis’ team studied four different MHC-I alleles and their interactions with molecular partners and antigens. Nuclear magnetic resonance (NMR) technology was used to reveal dynamic structural changes in MHC-1 protein. One of the functions of chaperones is to help MHC-1 proteins fold into their active shapes and stabilize them to prevent misfolding and aggregation. However, only some MHC-1 alleles rely on chaperone proteins for antigen loading. It still remains a mystery as to why some molecules rely on chaperone proteins, while others don’t. The researchers found that if the three-dimensional structure of the MHC-1 molecule is rigid, the chaperones are not involved in antigen loading. However, if it is flexible in the peptide-binding groove, the chaperone will interact with it and help the antigen loading process through ejecting antigens with low affinity to the binding groove, thereby ensuring that the MHC-1 protein binds only high-affinity antigens that can be displayed on the cell surface in the appropriate conformation, and thereby activating the T cell response.
By using flow cytometry and with the advance in fluorescently labeled tetrameric MHC–peptide complex, it has been made possible to directly visualize, quantify and characterize antigen-specific T cells, which has nearly transformed scientists’ understanding of cellular immune responses.
References
- McShan, A. C., Devlin, C. A., Overall, S. A., Park, J., Toor, J. S., Moschidi, D., … & Sgourakis, N. G. (2019). Molecular determinants of chaperone interactions on MHC-I for folding and antigen repertoire selection. Proceedings of the National Academy of Sciences.
Related Products