The FDA’s Center for Drug Evaluation and Research (CDER) regulatory approval of a total of 26 NME (New Molecular Entity) drugs in the first half of 2023. It is worth noting that the 26 drugs approved include 4 peptide drugs, accounting for 15% of the total.
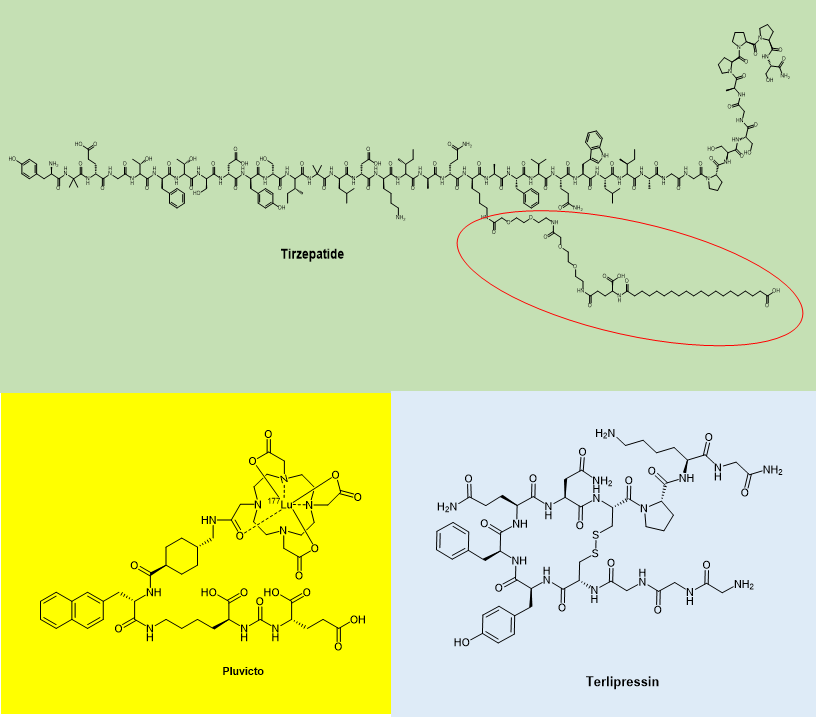
In the past 2022, the FDA has approved only three peptide drugs: Tirzepatide, Lutetium 177Lu Vipivotide Tetraxetan and Terlipressin, of which terlipressin was previously available outside the United States as Glypressin®. There is also literature that classifies Gadopiclenol approved in 2022 as a peptide drug, but this classification is not rigorous. Gadopiclenol contains only an amide bond, but it is not a condensation of two amino acids and should not be considered a peptide.
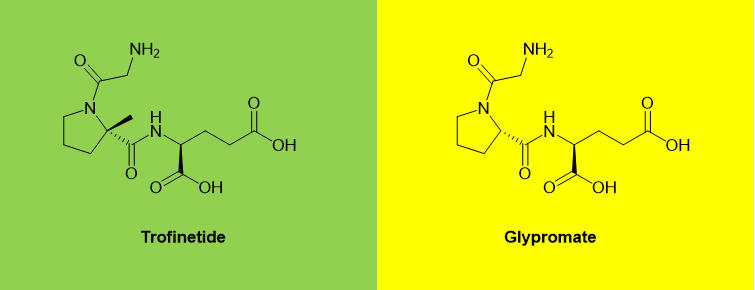
Trofinetide
Trofinetide is a novel synthetic analog of glypromate, also known as glycine-proline-glutamate (GPE). GPE is an N-terminal tripeptide of a naturally occurring protein and insulin-like growth factor 1 (IGF-1) in the brain. Compared with the proline Cα of GPE, trofinetide has one more methyl group.
Trofinetide (DaybueTM), developed by Acadia, is a peptide drug approved by the FDA for the treatment of Rett syndrome on March 10, 2023, making it the first FDA-approved Rett syndrome drug in history. Syndrome drugs. Rett syndrome is a rare disease with a spectrum of cognitive, motor, and autonomic symptoms. It is a complex, rare neurodevelopmental disorder usually caused by genetic mutations in the MECP2 gene. It is characterized by a period of normal development before 6 to 18 months of age, followed by significant developmental regression, with loss of acquired communication skills and use of the hands. Symptoms of Rett syndrome may also include hand rigidity, such as wringing and clapping, and abnormal gait. In the United States, Rett syndrome affects 6,000 to 9,000 patients, of whom approximately 4,500 have been diagnosed. The approval of Trofinetide is undoubtedly a milestone event in the history of human struggle against Rett syndrome.
- Mechanism of Action of Trofinetide
Most cases of Rett syndrome are associated with loss-of-function mutations in the gene encoding methyl CpG-binding protein 2 (MECP2). MECP2 is a DNA-binding protein that plays a role in the epigenetic regulation of gene expression. These mutations lead to immature synapses in the cerebral cortex, abnormal metabolism of cholesterol in the brain leading to abnormal neuronal development, and abnormal neuronal signaling. Rett syndrome is more common in girls than boys.
The exact mechanism of action of Trofinetide in Rett syndrome has not been fully elucidated. In mouse studies, GPE peptides improved exercise and cardiorespiratory function, increased brain weight, and extended the lifespan of MECP2-deficient mice. As a synthetic analogue of GPE, trofinetide similarly attenuates apoptosis and reduces infarct size in a dose-dependent manner in a rat model of hypoxic injury. Trofinetide plays a multifaceted role in reducing inflammation, excitotoxic-induced tissue damage and apoptosis, thereby protecting neurons and their surrounding underlying tissues.
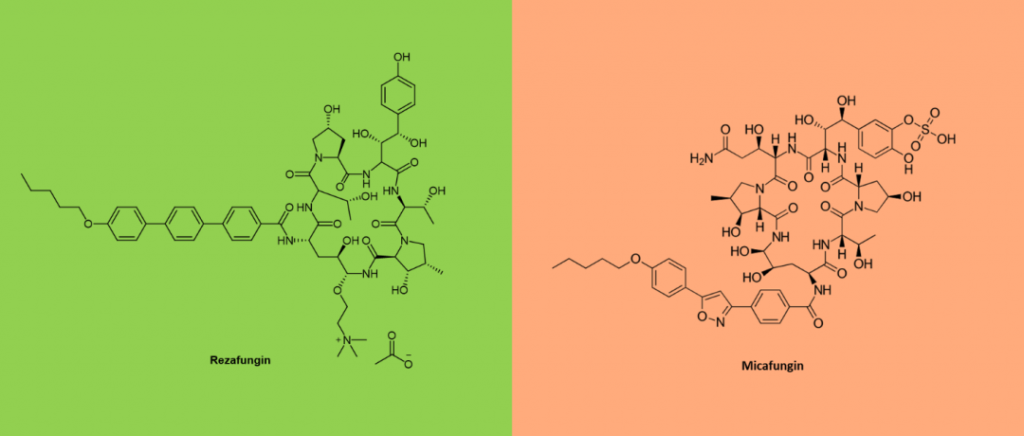
Rezafungin
New Echinocandin Peptide Drugs
On March 22, 2023, the FDA approved Cidara and Melinta’s RezzayoTM (rezafungin for injection) for the treatment of candidemia and invasive candidiasis in patients 18 years of age or older. Rezafungin is a new generation of broad-spectrum, long-acting echinocandin. Rezafungin has shown effective antifungal activity against Candida, Aspergillus and pneumospora sp. Rezafungin also became the first new echinocandin to receive FDA approval in more than a decade. Unlike other echinocinins such as caspofungin and micafungin, rezafungin has long-acting pharmacokinetics and high stability, allowing it to be administered at long intervals and maintain high plasma exposure. Rezafungin has a half-life of more than 130 hours and can be administered once a week instead of daily.
- Candidemia and Invasive Candidiasis
Candidemia and invasive candidiasis are rare, serious and life-threatening infections. According to the U.S. Centers for Disease Control and Prevention, the average rate of new infections in the United States is about 9 per 100,000 people, or about 25,000 cases per year. These infections are prone to occur in patients who already have other diseases and can lead to high morbidity and mortality. Patients at risk for invasive fungal infections include severely ill patients, immunosuppressed patients, patients after surgery, and patients using central venous catheters. In this already vulnerable patient population, invasive infections with candida often lead to serious illness and death. Patients with these invasive infections may develop a range of comorbidities on top of their underlying disease, including fever and septic shock. An additional hospital stay of 3 to 13 days is estimated after diagnosis of candidemia and invasive candidiasis. In addition, the mortality rate of these infected patients exceeds 40%. Echinocandins are used as first-line antifungals in practice guidelines for candidemia and invasive candidiasis because of their proven efficacy and safety as well as their powerful fungicidal activity.
- Another Cyclic Peptide
Rezafugin was discovered during the development of an orally administered Echinocin. Its outstanding structural feature is its chemical modification, mainly the addition of the choline moiety to the cyclic hexapeptide ring. This significantly improves molecular stability and biological properties. Compared to other echinocinins, the reduced metabolism and degradation of rezafungin enhances its stability and prolongates the half-life of the drug. This made it possible to administer the drug intravenously once a week.
- Mechanism of Action of Rezafungin
Rezafungin is an echinocandin antifungal drug. It inhibits the 1,3-β-D-glucan synthase complex in the fungal cell wall, which is responsible for the formation of 1,3-β-D-glucan. Since 1,3-β-D-glucan is an important component of fungal cell walls, rezafungin disrupts the cell walls of fungal species including Candida and acts as a concentration-dependent fungicide in vitro. Mammalian cells do not express β-1-3-glucan synthase, therefore, the action of rezafungin is specific to fungi.
Posluma (flotufolastat F 18)
Posluma (flotufolastat F 18) is an 18F-labeled ligand and an imaging agent for the detection and localization of prostate cancer in PET (positron emission tomography). Posluma contains an imaging agent (18F) whose active ingredient is flotufolastat F 18 gallium bound to non-radioactive gallium. Flotufolastat F 18 binds to prostate-specific membrane antigen (PSMA) (IC50= 4.4 nM) expressed on cells, including prostate cancer cells. In patients with recurrent prostate cancer who require local treatment, positron emission tomography (PET) with an 18F-labeled ligand provides accurate diagnostic imaging. The 18F group in Flotufolastat is a beta + emitting radionuclide that can be detected using PET. Since prostate cancer cells overexpress PSMA and flotufolastat F-18 is internalized by the cells, this radioactive agent can be used for diagnostic imaging in prostate cancer patients. Posluma received FDA regulatory approval on May 25, 2023 for PET of PSMA-positive lesions in men with suspected metastatic prostate cancer. It is the first FDA-approved PSMA-targeted imaging agent developed using a proprietary radiohybridization technology.
Paxlovid (nirmatrelvir, ritonavir)
The two active substances in Paxlovid, nirmatrelvir and ritonavir, are both peptidomimetics. Nirmatrelvir acts as a protease inhibitor to block the activity of proteases required for virus reproduction. It is an inhibitor of 3C-like protease (3C-like protease, 3CLPRO, also known as Mpro), which plays a key role in the replication of SARS-CoV-2 virus. The second peptidomimetic active substance ritonavir in Paxlovid is used as a pharmacokinetic enhancer of nirmatrelvir, which increases the half-life and bioavailability of nirmatrelvir by inhibiting the degradation of nirmatrelvir by CYP3A4.