Targeted Drug Delivery Peptide Therapeutics Development Cell-penetrating Peptide Conjugation Advanced Cyclization Techniques
Creative Peptides provides a wide range of cyclic peptide synthesis, which play a key role in different processes and have good therapeutic potential because they have several favorable properties, such as displaying a large surface area, which leads to high affinity and selectivity for protein targets, as well as their limited conformational flexibility, reducing entropy penalties during binding, thus improving their binding performance.
Depending on the cyclization position, there are several methods to synthesize cyclic peptides: head-to-tail, side-chain-to-side-chain, head-to-side-chain, and side-chain-to-tail, head-to-backbone, backbone-to-tail, side-chain-to-backbone, backbone-to-side-chain. (see figure below). While head-to-tail cycles are usually formed by amide bond formation, side-chain-to-side-chain cycles are most often synthesized via Cys-Cys or amide bond formation.
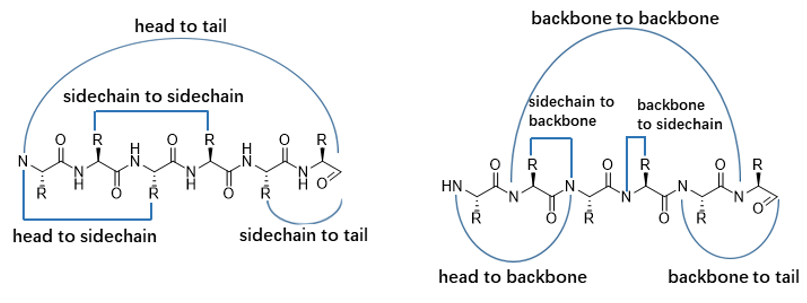
Backbone cyclic peptides are easily assembled via the formation of an amide bond between the peptides own N-terminus amine and C-terminus carboxylic acid. Our commonly used methods are chemical synthesis and biosynthesis. Creative Peptides perform the reaction with very high yields, and routinely provide over 98% purified cycled peptides.
Side chain-to-side chain cyclization is frequently employed to stabilize and select specific conformations and to reduce susceptibility toward proteolytic degradation. Creative Peptides have the capable to selectively protect, and then deprotect peptide sidechains to allow for the formation of lactam bridges between Lysine or Diaminopropanoic acid, and Glutamic and Aspartic Acids. Creative Peptides can even perform multiple different sites for bridge formation.
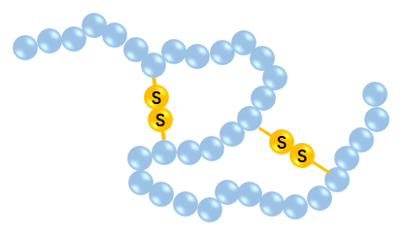
Disulfide bonds are widely present in hormones, enzymes, and immunoglobulins, and are thought to play an important role in reconstructing the conformation of biologically active peptides. To further enhance biological activity, people often introduce disulfide bonds to modify bioactive peptides. Creative Peptides usually add two cysteine residues to the peptide sequence, and the dilute solution of the peptide undergoes mild oxidation to form the desired intramolecular disulfide bond between the two cysteine residues. Ugi reaction also can be provided.
Disulfide bonds can be easily reduced to their acyclic mercaptan form in the intracellular environment. This challenge can be overcome by hydrocarbon-bound peptide synthesis, which can form a stable α-helix structure. It can mimic the typical molecular structure at the protein-protein interaction interface. When locked in this stable configuration, the constrained peptides can penetrate the cell and act on the protein targets in the cell.
Custom Cyclic Peptide Synthesis
We offer custom cyclic peptide synthesis for a variety of applications, from basic research to complex therapeutic peptides. Our team ensures high purity, efficiency, and flexibility in the synthesis process, enabling the production of peptides that meet your precise specifications.
High-Throughput Cyclic Peptide Synthesis
Accelerate your research with our high-throughput cyclic peptide synthesis services. We employ automated systems to produce large quantities of peptides quickly and efficiently, making it ideal for screening campaigns and large-scale projects.
Peptide Cyclization Techniques
Our peptide cyclization techniques include a range of methods for efficient ring formation, ensuring that the cyclic peptides are stable and functional. We use both chemical and enzymatic cyclization strategies to produce peptides with diverse ring structures.
Peptide Modification and Labeling
Enhance your cyclic peptides with various modifications such as fluorescent labeling, biotinylation, and PEGylation for improved bioavailability, stability, and detection. Our modification services are ideal for peptide-based assays and diagnostic applications
We provide cyclic peptide library construction services to help you explore diverse peptide sequences. These libraries are invaluable for drug discovery, biomarker identification, and functional screening.
Peptide Purification & Characterization
We use advanced techniques to purify and characterize your cyclic peptides to ensure they meet the highest standards. Our services include HPLC purification, mass spectrometry, and NMR analysis to confirm peptide identity, purity, and structure.
Peptide Therapeutics Development
Explore the therapeutic potential of cyclic peptides with our specialized services for drug development. From initial synthesis to preclinical studies, we assist in developing peptide-based therapeutics for cancer, autoimmune diseases, neurodegenerative diseases, and more.
Contract Research and Collaboration
Partner with us for contract research and collaborative projects. Whether you're a biotech company, pharmaceutical firm, or academic institution, we offer comprehensive support for your cyclic peptide research needs.
We specialize in TAT peptide synthesis, offering high-quality peptides for drug delivery, gene therapy, and targeted cellular interactions. Our expertise ensures efficient and reliable synthesis with optimal purity and bioactivity, ideal for your research and therapeutic applications.
DOTAGA Chelating Peptide Synthesis
Our DOTAGA chelating peptide synthesis service provides custom peptides with DOTAGA (1,4,7,10-Tetraazacyclododecane-N,N',N'',N'''-tetraacetic acid) for use in radiolabeling and molecular imaging. With precise synthesis techniques, we ensure high-quality peptides for applications in diagnostic imaging and targeted therapies.
Synthesis of Head-to-Tail Cyclic Peptides
We offer head-to-tail cyclic peptide synthesis, a method that forms stable cyclic peptides through reliable chemical or enzymatic cyclization techniques. Our service ensures robust peptide structures with enhanced stability, ideal for drug discovery and biomolecular research.
Our bicyclic peptides synthesis service delivers peptides with two interconnected rings, enhancing stability and bioactivity. By utilizing advanced cyclization techniques, we provide highly potent peptides suitable for therapeutic, diagnostic, and bioengineering applications.
Experienced Peptide Synthesis Experts
Our team of specialists brings years of expertise in cyclic peptide synthesis, ensuring high-quality results.
State-of-the-art Facilities and Equipment
We utilize the latest technology and equipment to deliver precise and reliable peptide synthesis.
Competitive Pricing
We offer cost-effective solutions without compromising on quality, making our services accessible for all project sizes.
Exceptional Quality Control and Testing
Every peptide we synthesize undergoes rigorous testing to guarantee purity and functionality.
Global Client Base with Proven Results
We have successfully delivered custom cyclic peptides to clients worldwide, ensuring high customer satisfaction and trust.
Scalability
From small-scale to high-throughput peptide synthesis, we provide flexible solutions for any project size.
1
Initial Consultation
We begin with a detailed consultation to understand your specific needs, including peptide sequence, modifications, and application requirements.
2
Custom Peptide Design
Our experts work with you to design a custom cyclic peptide that fits your research goals, ensuring the peptide sequence and cyclization method are optimized for stability and bioactivity.
3
Peptide Synthesis
Using solid-phase peptide synthesis (SPPS), we efficiently assemble your cyclic peptide, ensuring high purity and yield throughout the process.
4
Peptide Purification & QC
We employ HPLC purification and mass spectrometry (MS) to thoroughly purify and confirm the identity and structure of your cyclic peptide, ensuring it meets the highest quality standards.
5
Peptide Modification (if needed)
If necessary, we add functional groups such as fluorescent labeling, biotinylation, or PEGylation to enhance the peptide's functionality for applications like drug delivery or diagnostics.
6
Delivery & Support
Once your peptide is synthesized, purified, and modified, we provide fast delivery and ongoing support, ensuring it meets your research or development timelines.
Creative Peptides also has extensive experience in the design and production of modified peptides. We provide a variety of peptide modifications, labeling, and bioconjugation with other biomolecules, including
In addition, we also provide a variety of optional peptide analysis, including
Creative Peptides provides one-stop peptide solutions, from peptide synthesis, modification, labeling and conjugation to a wide range of biopolymers and surface attachments. Our peptides conjugation services include, but are not limited to
| CAT# | Product Name | M.W | Molecular Formula | Get a Quote |
| 10-101-103 | Vancomycin | 1449.25 | C66H75Cl2N9O24 | Inquiry |
| 10-101-104 | Teicoplanin | 1879.66 | C88H95Cl2N9O33 | Inquiry |
| 10-101-169 | Pasireotide | 1047.2 | C58H66N10O9 | Inquiry |
| 10-101-186 | Romidepsin | 540.69584 | C24H36N4O6S2 | Inquiry |
| 10-101-325 | Semaglutide | 4113.57 | C187H291N45O59 | Inquiry |
| 10-101-62 | Ziconotide | 2639.2 | C102H172N36O32S7 | Inquiry |
| 10-101-78 | Dalbavancin | 1816.69 | C88H100Cl2N10O28 | Inquiry |
| AF083 | Polymyxin B | Inquiry | ||
| MFP-041 | Rezafungin | 1226.4 | C63H85N8O17 | Inquiry |
| R04030 | Cyclo(-Arg-Gly-Asp-D-Phe-Val) | 574.6 | C26H38N8O7 | Inquiry |
| R1574 | Octreotide | 1019.2 | C49H66N10O10S2 | Inquiry |
| R1812 | Lanreotide | 1096.3 | C54H69N11O10S2 | Inquiry |
| R1824 | Cyclo(RGDyK) | 847.7 | C31H43F6N9O12 | Inquiry |
| R2018 | Capreomycin | Inquiry | ||
| R2029 | Enviomycin | 685.69 | C26H43N11O11 | Inquiry |
| R2052 | Zilucoplan | 3562 | C172H278N24O55 | Inquiry |
| R2238 | Telavancin | 1755.6 | C80H106Cl2N11O27P | Inquiry |
| R2239 | Oritavancin | 1793.1 | C86H97Cl3N10O26 | Inquiry |
| R2240 | Bacitracin | 1422.69 | C66H103N17O16S | Inquiry |
| R2241 | Viomycin | 685.69 | C25H43N13O10 | Inquiry |
| R2242 | Colistin | 1169.5 | C53H100N16O13 (for E1) | Inquiry |
| R2243 | Micafungin | 1270.27 | C56H71N9O23S | Inquiry |
| R2244 | Anidulafungin | 1140.24 | C58H73N7O17 | Inquiry |
| R2245 | Fostamatinib | 580.46 | C23H26FN6O9P | Inquiry |
| R2246 | Paritaprevir | 765.88 | C40H43N7O7S | Inquiry |
| R2247 | Grazoprevir | 766.9 | C38H50N6O9S | Inquiry |
| Z10-101-154 | Daptomycin | 1620.69 | C72H101N17O26 | Inquiry |
| Z10-101-157 | Caspofungin | 1093.31 | C52H88N10O15 | Inquiry |
Cyclic peptide synthesis is the process of creating peptides that form a ring structure, which enhances their stability, bioactivity, and specificity. This technique is widely used for applications in drug discovery, cancer therapy, diagnostic imaging, and more.
Cyclic peptides offer enhanced stability compared to linear peptides, making them more effective for therapeutic use. Their unique structure also allows for higher affinity binding to target molecules, which is especially beneficial in drug discovery, cancer therapy, and diagnostic imaging.
Yes, we offer fully customizable cyclic peptide synthesis. Whether you need peptides for drug development, targeted drug delivery, or biomolecular research, we tailor the synthesis process to meet your exact needs, including modifications like fluorescent labeling, biotinylation, and PEGylation.
We employ rigorous quality control measures, including HPLC purification, mass spectrometry, and NMR analysis to verify the identity, purity, and structure of each peptide. This ensures that our cyclic peptides meet the highest standards for scientific and therapeutic use.
Absolutely! We provide high-throughput cyclic peptide synthesis services to accommodate large-scale production needs, ideal for screening campaigns, peptide libraries, and extensive research studies. Our automated systems ensure consistent quality and rapid delivery for large quantities.
