PEGylated Peptide DevelopmentSite-specific PEGylationPEG Linker ModificationPeptide Pharmacokinetics Optimization
At Creative Peptides, we specialize in custom peptide PEGylation services designed to improve the pharmacokinetic performance, stability, and therapeutic potential of peptide-based drugs and biologics. PEGylation — the covalent attachment of polyethylene glycol (PEG) chains to peptides — is a well-established strategy widely used in pharmaceutical development to enhance solubility, extend circulation half-life, reduce proteolytic degradation, and minimize immunogenicity.
Our team integrates advanced peptide synthesis, site-specific PEG conjugation technologies, and rigorous analytical characterization to deliver highly controlled PEGylated peptide products tailored for research, preclinical development, and GMP manufacturing. Whether you are optimizing therapeutic peptides, improving peptide drug delivery, or developing long-acting peptide biologics, our PEGylation platform provides reliable and scalable solutions that meet the strict quality expectations of biotechnology and pharmaceutical enterprises.
Although peptide therapeutics offer high biological specificity and strong target affinity, their clinical translation often faces major challenges such as rapid enzymatic degradation, short plasma half-life, low solubility, and frequent dosing requirements. These limitations can reduce therapeutic efficacy and increase development risks for peptide drug candidates.
Peptide PEGylation has become a widely adopted modification strategy in the biopharmaceutical industry because it addresses these critical barriers by:
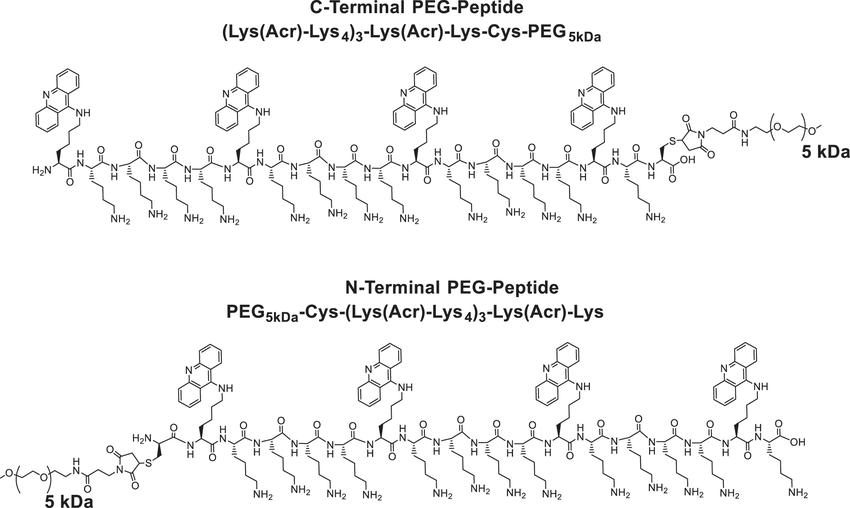 Fig. 1. Structure of PEGylated polyacridine peptides (Gene therapy. 2020, 27(5): 196-208).
Fig. 1. Structure of PEGylated polyacridine peptides (Gene therapy. 2020, 27(5): 196-208).
Creative Peptides provides integrated peptide PEGylation services supporting pharmaceutical companies, biotechnology firms, and academic research organizations developing peptide therapeutics. Our PEGylation platform combines advanced peptide synthesis technologies, diverse PEG reagent libraries, site-specific conjugation chemistries, and rigorous analytical validation to ensure reliable modification outcomes.
Our services are designed to support peptide drug discovery, pharmacokinetic optimization, and preclinical development programs. Each project is customized according to peptide sequence characteristics, therapeutic goals, PEG architecture, and regulatory development requirements.
Successful peptide PEGylation begins with a rational modification strategy. Our scientists collaborate with clients to evaluate peptide structure, functional regions, and pharmacokinetic objectives in order to design an optimal PEGylation approach.
This strategic planning stage ensures that PEGylation improves pharmacokinetics while preserving peptide bioactivity and receptor binding.
We provide high-quality peptide synthesis optimized for downstream PEGylation reactions. Our peptide platform utilizes automated solid-phase peptide synthesis (SPPS) combined with advanced purification and characterization technologies.
Our synthesis workflow ensures high peptide purity and consistent modification compatibility prior to PEG conjugation.
The choice of PEG reagent strongly influences conjugation efficiency and therapeutic performance. We support projects using a wide variety of functional PEG reagents commonly applied in peptide drug development.
We assist clients in selecting PEG structures that balance stability, activity retention, and manufacturability.
Our PEGylation platform supports multiple conjugation chemistries that enable precise modification while preserving peptide structure and biological function.
Reaction conditions are carefully optimized to maximize PEGylation efficiency and maintain peptide integrity.
PEGylated peptides require advanced analytical validation due to increased molecular heterogeneity and structural complexity. Our analytical platform ensures accurate characterization and quality control.
For peptide drug candidates advancing toward preclinical or clinical development, scalable PEGylation processes are essential. Our team supports process optimization and manufacturing scale-up to ensure reproducibility and regulatory readiness.
These services help accelerate the transition from discovery-stage peptide modification to large-scale therapeutic development.
To further support peptide drug development programs, we can coordinate stability and pharmacokinetic testing through specialized partner laboratories.
These studies provide critical data for evaluating the therapeutic benefits of PEGylation during early-stage development.
Enterprise peptide programs typically choose PEGylation strategies based on three decision drivers: (1) control of conjugation site to preserve bioactivity, (2) product homogeneity for reproducible CMC, and (3) scalability of the chemistry and purification. The table below summarizes commonly deployed PEGylation approaches in peptide and peptide-like biologics development.
| Strategy | Preferred Conjugation Site | Typical Activated PEG Chemistry | Key Advantages (Enterprise Focus) | Typical Use Cases |
|---|---|---|---|---|
| N-terminal PEGylation (site-preferred) | N-terminus (α-amine) | Aldehyde-PEG (reductive amination) or other N-terminal selective methods | Often improves site control vs. lysine targeting; can help preserve internal pharmacophores | Half-life extension where receptor-binding region must remain unobstructed |
| Cysteine-selective PEGylation | Free cysteine (native or engineered) | Maleimide-PEG (thiol coupling) | High site selectivity; supports more homogeneous products and cleaner impurity profiles | Controlled mono-PEGylation; programs requiring strong lot-to-lot consistency |
| Lysine-directed PEGylation | Lysine ε-amine and/or N-terminus | NHS-PEG (amine coupling) | Broad applicability and operational simplicity; useful for early feasibility work | Screening studies; candidates tolerant to some heterogeneity |
| Bioorthogonal "Click" PEGylation | Engineered azide/alkyne handle | Azide-PEG / Alkyne-PEG (click chemistry routes) | Strong site control; can reduce side reactions; compatible with defined conjugation designs | Programs needing precise positioning for activity retention and IP positioning |
| Cleavable (releasable) PEGylation | Defined site (depends on linker design) | Activated PEG bearing cleavable linker motifs | Balances PK extension with potential restoration of native activity at target site | Delivery-focused programs; candidates sensitive to steric shielding |
PEG architecture selection is a high-impact design choice for peptide half-life extension, solubility improvement, and manufacturability. In outsourced development, enterprise teams typically evaluate architecture alongside PEG molecular weight, linker stability, and the risk of activity loss due to steric shielding.
| PEG Type | Structural Characteristics | Common Selection Rationale | Practical Considerations | Typical Applications |
|---|---|---|---|---|
| Linear PEG | Single-chain PEG with one reactive end (or two for bifunctional designs) | Widely used baseline option for PK and solubility improvements | Can reduce activity if attached near binding epitope; easier to manufacture and characterize | General half-life extension; formulation support |
| Branched PEG | Two-chain (Y-shaped) or branched configuration | Greater hydrodynamic size at similar PEG mass to extend systemic exposure | May increase steric shielding; purification/analytics can be more complex than linear PEG | Long-acting designs requiring stronger renal clearance reduction |
| Multi-arm PEG | 3–8 arm PEG structures with multiple termini | Used for advanced constructs or multi-functional designs | Higher complexity; careful control needed to avoid multi-attachment heterogeneity | Specialty delivery systems; scaffold-like conjugates |
| Heterobifunctional PEG | Two different reactive ends for stepwise conjugation | Enables controlled assembly and conjugate orientation | Requires tighter process control; useful for modular build strategies | Platform development; staged conjugations |
| Cleavable PEG (releasable) | PEG with linker designed to cleave under defined conditions | Designed to mitigate activity loss from PEG shielding while maintaining PK benefits | Linker stability testing becomes critical; needs application-aligned release rationale | Activity-sensitive peptides; delivery-focused therapeutic concepts |
Activated PEG selection is driven by peptide functional groups, desired site selectivity, and impurity control requirements. This table summarizes common functional PEG chemistries used in peptide PEGylation programs, supporting both early feasibility work and more controlled, development-oriented workflows.
| Functional PEG Reagent | Reactive Group | Primary Target on Peptide | Typical Chemistry | Notes for Development Programs |
|---|---|---|---|---|
| NHS-PEG | N-hydroxysuccinimide ester | Primary amines (lysine ε-amine, N-terminus) | Amide bond formation | Operationally simple but can increase heterogeneity if multiple lysines are present |
| Maleimide-PEG | Maleimide | Free thiol (cysteine) | Thiol addition (selective coupling) | Often preferred for site-specific mono-PEGylation when a unique cysteine is available |
| Aldehyde-PEG | Aldehyde | N-terminus (α-amine) | Reductive amination | Common approach for N-terminal bias; supports improved control vs. broad lysine targeting |
| Azide-PEG | Azide | Alkyne handle (engineered) | Click chemistry routes | Strong site control; requires installation of compatible handle on peptide |
| Alkyne-PEG | Alkyne | Azide handle (engineered) | Click chemistry routes | Often paired with azide-bearing peptides for defined conjugation |
| Product (INN / Brand) | Molecule Type | PEGylation Note | Therapeutic Area | Market Status (High-Level) |
|---|---|---|---|---|
| Pegvisomant / Somavert | Pegylated recombinant GH analogue (protein/peptide-like biologic) | Pegvisomant contains covalently bound polyethylene glycol (PEG) polymers (multiple PEGs per molecule reported in labeling) | Acromegaly | Authorized in EU / Approved in US labeling describes PEG conjugation :contentReference[oaicite:0]{index=0} |
| Pegfilgrastim / Neulasta | Pegylated protein (G-CSF) | Filgrastim is "pegylated" with polyethylene glycol to slow clearance | Neutropenia support during chemotherapy | EU authorized (example of clinically validated PEGylation) :contentReference[oaicite:1]{index=1} |
| Certolizumab pegol / Cimzia | PEGylated antibody fragment (Fab') | PEGylated Fab' fragment used to extend exposure | Immune-mediated inflammatory diseases | EU authorized product information available :contentReference[oaicite:2]{index=2} |
| Peginesatide / Omontys | PEGylated peptide (synthetic peptide ESA) | Withdrawn following serious hypersensitivity/anaphylaxis reports (post-marketing) | Anemia in CKD patients on dialysis | US approval withdrawn after recall (regulatory record) :contentReference[oaicite:3]{index=3} |
Specialized Peptide Modification Expertise
Our team has extensive experience in peptide chemistry and bioconjugation, enabling the design of PEGylation strategies that preserve peptide bioactivity while improving pharmacokinetic performance.
Multiple PEGylation Technologies
We support diverse PEGylation approaches including N-terminal modification, cysteine-selective PEGylation, lysine-directed conjugation, and bioorthogonal conjugation strategies.
Broad PEG Reagent Selection
Our platform supports a wide range of PEG architectures including linear PEG, branched PEG, and functionalized PEG derivatives commonly used in peptide therapeutic development.
Controlled Conjugation Strategies
Optimized conjugation conditions enable controlled PEGylation and help reduce heterogeneous product mixtures during peptide modification.
Comprehensive Analytical Support
Each PEGylated peptide product is characterized using advanced analytical methods including LC-MS, HPLC, and other orthogonal techniques to confirm identity and purity.
Support for Drug Development Programs
Our services support multiple stages of peptide therapeutic development, from feasibility studies and lead optimization to process development for larger-scale production.
Flexible Project Design
PEGylation projects can be customized according to peptide structure, desired pharmacokinetic profile, and specific research objectives.
Reliable Technical Communication
Our scientists work closely with enterprise clients to ensure transparent communication, technical clarity, and consistent project progress.
Integrated Peptide Development Capabilities
In addition to PEGylation, we support peptide synthesis, modification, and analytical services that help streamline peptide drug research workflows.
Peptide PEGylation projects require careful coordination between molecular design, conjugation chemistry, and analytical verification. Our workflow is designed to provide enterprise clients with a structured and traceable development process that supports early feasibility studies as well as later-stage drug development programs.
1
Technical Consultation & PEGylation Strategy Design
2
Peptide Preparation & Functionalization
3
PEGylation Reaction Development
4
Purification & Analytical Characterization
5
Process Optimization & Project Delivery
Peptide PEGylation is widely used in pharmaceutical research and biotechnology to improve the pharmacokinetic and physicochemical properties of peptide-based molecules. By increasing hydrodynamic size and shielding peptides from enzymatic degradation, PEGylation can enhance systemic exposure, improve formulation stability, and support the development of long-acting peptide therapeutics. The following sections summarize common application areas where peptide PEGylation strategies are applied.
If you are developing peptide therapeutics, optimizing peptide stability, or exploring pharmacokinetic enhancement strategies, peptide PEGylation may provide an effective modification approach. Creative Peptides offers customized peptide PEGylation services supported by experienced peptide chemists, diverse PEG reagent options, and advanced analytical capabilities. Our team works closely with biotechnology companies, pharmaceutical developers, and academic researchers to design PEGylation strategies that align with project objectives and development timelines. From early feasibility studies to scalable process development, we provide technical support throughout the peptide modification workflow.
Contact our scientists today to discuss your peptide PEGylation project or request a consultation.