Label-Free Interaction AnalysisMultiplex Binding StudiesBiochip Design & PrintingKinetics and Affinity Profiling
At Creative Peptides, we provide custom Surface Plasmon Resonance Imaging (SPRi) services for research teams that need real-time, label-free, and multiplex interaction analysis with practical experimental support. Our SPRi platform is suited to peptide, protein, antibody, nucleic acid, carbohydrate, and other biomolecular binding studies where throughput, chip-level parallelism, and data comparability matter. By combining assay planning, biochip preparation, sample immobilization strategy, interaction testing, and data interpretation, we help clients move from feasibility questions to decision-useful binding data with workflows tailored to screening, characterization, and assay development. For projects that require matched reagents, we can also integrate upstream support in custom peptide synthesis, custom peptide labeling, and biotinylated peptides.
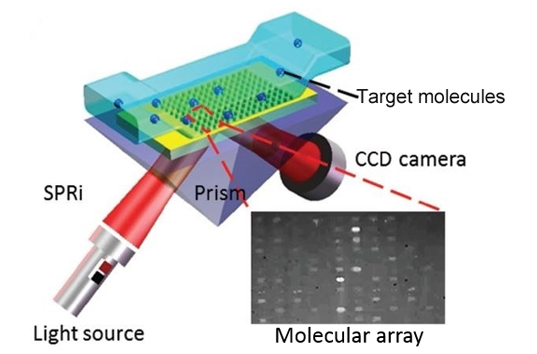
Many interaction studies become inefficient when teams must choose between throughput and data richness. Conventional one-by-one testing can slow candidate ranking, consume too much sample, and make it difficult to compare multiple ligands, immobilization conditions, or analyte concentrations within a consistent experimental frame.
SPRi helps address these practical challenges by:
We offer flexible SPRi workflows for clients who need more than instrument access alone. Projects can be built around client-supplied materials or integrated with our upstream peptide and assay-support capabilities, including peptide library and array preparation and peptide array-based epitope mapping when sequence-level screening strategies are required.
Effective SPRi studies start with a clear assay plan. We review target type, ligand and analyte format, expected binding strength, matrix complexity, required controls, and the intended decision point before selecting a practical experimental route.
This front-end design step helps reduce avoidable rework and improves the likelihood of generating interpretable data from the first study cycle.
Biochip configuration has a direct effect on assay robustness and data consistency. We support chip-level setup for multiplex SPRi experiments with layouts designed around the study question rather than a fixed template.
This service is particularly useful for clients who need array-style study design but do not want to manage chip preparation in-house.
SPRi is widely used to monitor association and dissociation behavior in real time while comparing multiple interactions in parallel. We support assay execution and data review for projects that need more than endpoint binding confirmation.
Our goal is to provide clean, decision-supportive data packages that help clients compare candidates with greater confidence.
SPRi is especially valuable when the project requires broad interaction coverage rather than a narrow single-format assay. We support a range of biomolecular interaction models relevant to discovery and analytical development.
When a project requires matched biology tools, we can connect SPRi work with antigen-antibody interaction service and epitope mapping services.
Many clients choose SPRi because they need to compare multiple candidates, conditions, or binding relationships on a single platform. We design screening workflows that make this advantage operational rather than theoretical.
These workflows are suited to programs that need broad screening output before selecting a smaller set for deeper follow-up work.
Reliable SPRi data depends heavily on reagent quality, orientation, and compatibility with the selected surface strategy. We support reagent preparation to improve assay readiness and experimental flexibility.
For projects requiring modified assay materials, our team can integrate fluorescence and dye-labeled peptide services and peptide modification services into the SPRi workflow.
SPRi projects generate more value when the output is organized around the client's decision needs. We provide reporting that focuses on interpretation as well as raw signal capture.
Different SPRi projects are driven by different decision questions. The table below summarizes common study types and the practical value they provide during assay development and interaction screening.
| Study Type | Main Objective | Typical Samples | Representative Output | Why Clients Request It |
|---|---|---|---|---|
| Affinity Screening | Rank binders across a panel in a consistent format | Peptides, antibodies, proteins, nucleic acids | Relative binding strength, response comparison, hit ranking | Faster triage of promising candidates before deeper characterization |
| Kinetic Profiling | Examine association and dissociation behavior in real time | Protein-protein, peptide-protein, antibody-antigen systems | Sensorgrams, kinetic trend analysis, fitted interaction models | Better understanding of binding quality beyond endpoint signal |
| Multiplex Ligand Comparison | Compare multiple immobilized ligands on one chip | Peptide panels, sequence variants, capture reagents | Side-by-side response maps and comparative binding behavior | More efficient screening with reduced run-to-run variability |
| Competition / Blocking Analysis | Determine whether binding events overlap or interfere | Antibodies, peptides, receptors, antigens | Competition patterns, blocking relationships, comparative response shifts | Supports epitope-related assessment and assay mechanism clarification |
| Surface Chemistry Evaluation | Identify a practical immobilization and capture strategy | Immobilization-ready peptides, proteins, conjugates | Spot quality, signal consistency, background assessment | Reduces failure risk before scaling a broader study |
| Specificity / Cross-Reactivity Review | Check whether related analytes behave similarly or differently | Variant peptides, homologous proteins, control panels | Comparative response patterns and selectivity trends | Helps refine candidate selection and assay confidence |
SPRi performance depends not only on the instrument, but also on how the study is configured. The following table highlights common design variables that influence data quality and project fit.
| Design Factor | What Needs to Be Decided | Typical Options | Impact on Results | Development Consideration |
|---|---|---|---|---|
| Ligand Immobilization Format | Which binding partner should be placed on the chip? | Direct spotting, capture-based immobilization, biotin-streptavidin, covalent coupling | Influences orientation, accessibility, and reproducibility | Choice should balance stability with preservation of binding function |
| Chip Layout | How should samples, controls, and replicates be arranged? | Candidate panels, concentration series, reference spots, blank controls | Affects data comparability and screening efficiency | Layout should align with the final decision question, not just chip capacity |
| Buffer and Matrix Conditions | What running conditions best support stable signal? | Standard assay buffers, detergent-containing buffers, salt or pH variants | Can alter nonspecific binding, baseline behavior, and regeneration success | Early feasibility work often prevents avoidable interpretation problems later |
| Analyte Concentration Strategy | Should the study focus on screening, ranking, or model fitting? | Single concentration, concentration series, titration sets | Determines whether output is qualitative, comparative, or kinetic | Sample availability often guides the most practical design |
| Regeneration Approach | Can the chip surface be reused without damaging performance? | Mild regeneration, no-regeneration workflows, capture-refresh formats | Affects throughput, surface stability, and cycle-to-cycle consistency | Regeneration needs to be tested against ligand robustness and assay objective |
| Readout Priority | What is the primary decision metric? | Hit calling, affinity ranking, kinetics, competition, specificity | Shapes assay design and downstream analysis method | Clear endpoint definition improves reporting and study efficiency |
Multiplex Study Design
We build chip layouts around the real comparison the client needs to make, helping convert array capacity into usable project value.
Label-Free Readout
Real-time monitoring supports direct interaction analysis without relying on secondary labels or indirect signal systems.
Flexible Molecule Coverage
Our SPRi workflows can be configured for peptides, proteins, antibodies, nucleic acids, carbohydrates, and related biomolecular systems.
Chip and Reagent Support
We support both assay execution and the upstream preparation of immobilization-ready reagents and array-compatible materials.
Decision-Oriented Reporting
Results are organized to help clients compare candidates, assess assay quality, and identify the most practical next step.
Integrated Peptide Expertise
For peptide-centered projects, we can combine SPRi analysis with synthesis, labeling, modification, and panel design support in one workflow.
Our workflow is designed to move efficiently from study design to delivery of structured SPRi data for screening, characterization, and assay optimization.
1
Project Review and Assay Scoping
2
Reagent Preparation and Biochip Setup
3
SPRi Assay Execution
4
Data Processing and Quality Review
5
Reporting and Follow-On Recommendations
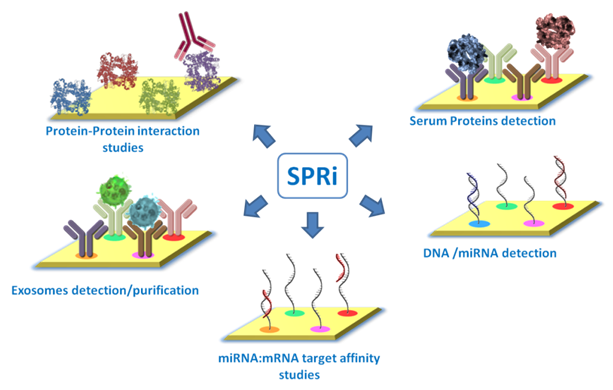
SPRi is valuable in programs where parallel interaction analysis can improve speed, comparability, and assay confidence. Below are representative use scenarios in which SPRi services can provide clear technical value.
SPRi relies on the principle of surface plasmon resonance, where changes in refractive index on a sensor surface, due to molecular binding events, alter the resonance condition of incident light. This change is detected as a shift in the SPR angle, providing quantitative information about binding kinetics and affinity.
SPRi can be used to investigate a wide range of interactions, including protein-protein, protein-DNA/RNA, protein-small molecule, antibody-antigen, and receptor-ligand interactions.
SPRi offers several advantages, including label-free detection, real-time monitoring of interactions, high sensitivity, minimal sample consumption, and the ability to study complex biological samples such as serum, cell lysates, and crude extracts.
We can analyze various types of samples, including purified proteins, antibodies, nucleic acids, small molecules, peptides, and complex biological samples.
Our SPRi data analysis provides comprehensive information, including kinetic parameters (association rate constant, dissociation rate constant, equilibrium dissociation constant), binding affinity, stoichiometry, and thermodynamic parameters (enthalpy, entropy).
The turnaround time for SPRi analysis depends on the complexity of the project and sample availability. We strive to deliver results in a timely manner and can discuss specific timelines based on your requirements.
Yes, our team of experts can assist you with experimental design, optimization of assay conditions, data interpretation, and recommendations for further experiments based on the results obtained from SPRi analysis.
Yes, our SPRi platform is capable of handling high-throughput screening projects, enabling rapid screening of compound libraries or protein interactions.

If your team needs a practical partner for Surface Plasmon Resonance Imaging studies, Creative Peptides can support your project with biochip design, multiplex assay planning, interaction testing, and data interpretation tailored to your research goals. Whether you are evaluating peptide binders, comparing antibodies, optimizing a surface strategy, or building a broader interaction screening workflow, we can help you generate clearer binding data with a service model built around real project needs. Contact us today to discuss your samples, assay objective, and SPRi study scope.