Protein-protein interactions (PPIs) are highly specific physical contacts established between two or more protein molecules, which are the result of biochemical events caused by electrostatic forces. It is critical to understand how they interact at residual levels during the early stage and later stage of drug development. Surface Plasmon Resonance imaging (SPRi), namely Surface Plasmon Resonance Microscopy (SPRM), is a high-sensitive, real-time, label-free, and high-throughput technique which is used to study biomolecular interactions based on detecting the refractive index changes resulting from molecular binding. Moreover, protein-protein interactions play an important role in transcription and other biological processes.
Service Content: The affinities of candidate drug Antibody-1 and candidate drug Antibody-2 for EGFR, FcRn, FcγRIIIA were determined.
Methods: Multi-concentration gradient fitting, multiple 3-fold SD screening
Results: The reliable affinity constants were obtained for later biological experiments and clinical trials. The Application of SPRi technology significantly reduces the time and cost in protein-protein interaction assay.
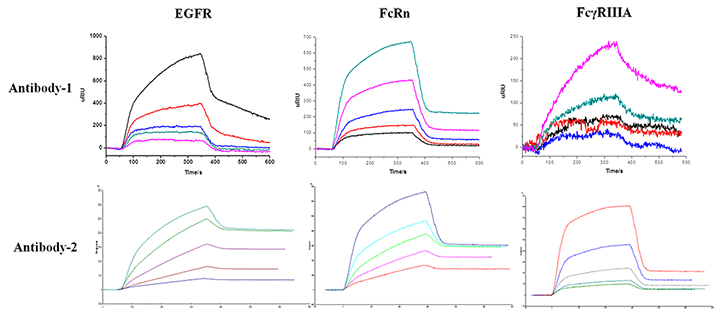
In conclusion, SPRi is a well-established leading technology for measuring binding association (ka) and dissociation rates (kd), affinities (KD) for protein-protein interactions.
Creative Peptides offers SPRi (Surface Plasmon Resonance imaging) services including Biochip design and printing, Bio-interactions analysis (binding affinity and kinetic processes detection), Summary and analysis of the results. The SPRi technology also shows great promise to study even more complex protein-protein interactions. We can follow the customer's request for high-precision analysis of protein-protein interactions while ensuring reasonable price and accurate analytical data.
Protein-protein interactions (PPIs) are crucial for various biological processes, including signal transduction, gene expression regulation, and enzymatic activities. They are specific physical contacts formed between two or more proteins, driven by biochemical forces such as electrostatic interactions.
Surface Plasmon Resonance imaging (SPRi) is a powerful, real-time, label-free technique that monitors protein-protein interactions by detecting changes in the refractive index caused by molecular binding. SPRi can measure binding affinity, kinetics, and dissociation rates, providing precise insights into protein interaction dynamics.
SPRi is used to study protein-protein interactions in various biological contexts, such as drug development and disease research. It helps quantify binding affinities, determine kinetic parameters, and monitor complex protein interactions, making it invaluable for understanding molecular mechanisms and optimizing therapeutic strategies.
Creative Peptides offers SPRi services, including biochip design, interaction analysis, and result interpretation. We specialize in measuring binding affinities, kinetic processes, and providing detailed analytical reports. Our services are tailored to meet customer needs, offering reliable and cost-effective solutions for protein-protein interaction research.
Creative Peptides provides advanced SPRi technology, experienced scientific support, and high-quality results. We ensure reproducibility, accurate data analysis, and comprehensive reports to help accelerate your research and reduce costs in protein-protein interaction studies.

References