Site-specific Fluorescent LabelingClick Chemistry Peptide LabelingFor Imaging and Binding AssaysBiodistribution Studies
At Creative Peptides, we provide enterprise-grade fluorescence and dye labeling peptide services designed for advanced biological imaging, assay development, diagnostics, and translational research. By integrating high-purity peptide synthesis with site-specific fluorescent and chromogenic labeling strategies, we deliver robust, reproducible peptide probes that meet the stringent performance and quality requirements of pharmaceutical companies, biotechnology firms, CROs, and academic research institutions. Our expertise spans a broad spectrum of organic dyes and fluorophores, enabling precise signal control, high labeling efficiency, and compatibility with both in vitro and in vivo applications.
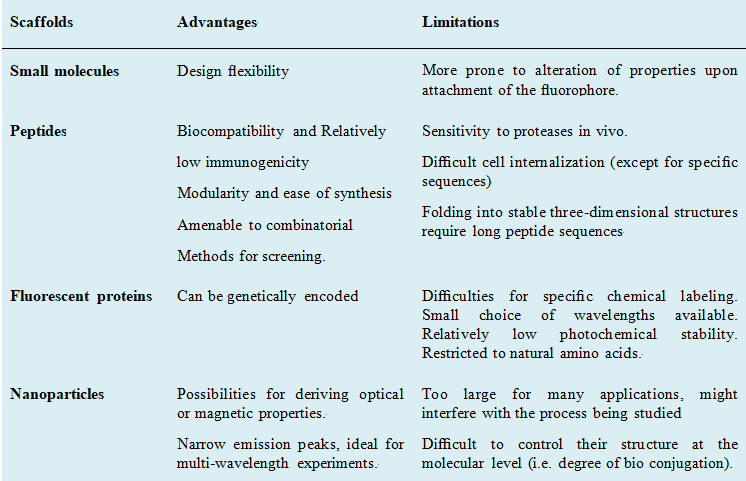 Advantages and limitations of different fluorescent probes.
Advantages and limitations of different fluorescent probes.
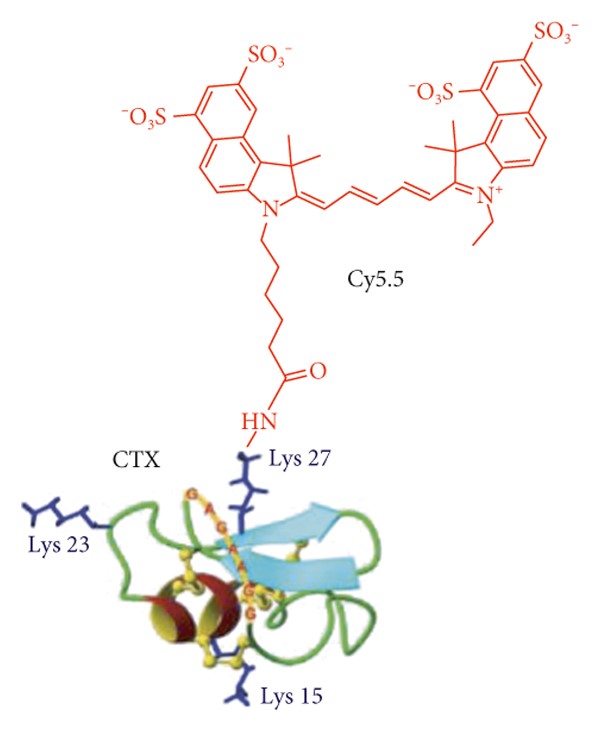 Peptide chlorotoxin (CTX) is labeled with Cy5.5. (Joshi, B. P., 2018)
Peptide chlorotoxin (CTX) is labeled with Cy5.5. (Joshi, B. P., 2018)Fluorescence and dye-labeled peptides are essential tools in modern life science research, yet conventional labeling approaches often suffer from inconsistent labeling efficiency, signal quenching, batch-to-batch variability, and limited biological compatibility.
Professional peptide fluorescence labeling directly addresses these challenges by:
Fluorescence and dye-labeled peptides are widely used across drug discovery, diagnostics, and bioanalytical research, yet many organizations face recurring challenges such as inconsistent labeling efficiency, signal instability, loss of biological activity, and limited scalability. Creative Peptides addresses these issues through a scientifically grounded, highly customizable service model that integrates advanced labeling chemistries, flexible peptide design, and close technical collaboration. Our services are structured to support enterprise customers requiring reliable data, reproducible performance, and long-term project continuity.
Selecting an appropriate fluorescent dye is not a catalog choice but a technical decision that directly impacts signal quality, assay robustness, and biological relevance. Our scientists work with you to define a fluorophore strategy based on experimental objectives, detection platforms, and downstream use.
This application-driven approach helps enterprise users shorten development timelines and improve data reliability across studies.
Fluorescent labeling can compromise peptide function if not carefully designed. We focus on preserving biological activity through rational peptide engineering and controlled conjugation strategies tailored to each sequence and application.
This flexibility allows clients to adapt labeling strategies as assays, targets, or readouts evolve, without rebuilding the entire peptide system.
Inconsistent labeling quality is a major source of irreproducible results. We implement multi-level analytical verification to ensure that labeled peptides meet defined performance and consistency criteria.
Each batch is accompanied by clear analytical documentation to support enterprise traceability, internal review, and study reproducibility.
Beyond synthesis and labeling, our service model emphasizes collaboration and technical continuity. Dedicated scientific support is provided throughout the project lifecycle.
This collaborative approach helps enterprise teams reduce development risk and maintain consistency across multi-phase research programs.
Fluorophore selection is a foundational decision in fluorescence-labeled peptide design. Different dye classes exhibit distinct spectral properties, stability profiles, and biological compatibility, which directly influence imaging quality, assay robustness, and experimental reproducibility. The table below summarizes commonly used fluorophore categories and how they are selected in enterprise research environments.
| Fluorophore Type | Spectral Region | Typical Enterprise Use Cases | Selection Rationale (Professional) |
|---|---|---|---|
| Fluorescein / Rhodamine Class | Visible (green/orange-red) | Microscopy labeling, flow cytometry method development, ligand binding studies | Widely used and instrument-compatible; selection depends on photostability needs and background conditions |
| Alexa Fluor-type / High-Photostability Dyes | Visible to far-red | High-content imaging, confocal microscopy, quantitative imaging workflows | Preferred when stronger photostability and consistent fluorescence output are required for long acquisition times |
| Cyanine (Cy) Dyes | Visible to near-infrared | Flow cytometry panels, multiplex assays, plate reader workflows | Common in multi-color workflows; requires careful planning for spectral overlap and dye proximity effects |
| Near-Infrared (NIR) Dyes | NIR | In vivo imaging feasibility studies, tissue imaging, low-autofluorescence applications | Chosen to reduce biological autofluorescence and improve imaging depth; stability and formulation considerations are important |
| Environment-Sensitive / Functional Dyes | Varies | Membrane interaction studies, uptake tracking, microenvironment-sensitive readouts | Used when fluorescence response depends on environment; requires validation under real assay conditions |
| > Fluorescein Isothiocyanate | > Anthranilyl | > 5/6-Carboxyfluorescein |
| > Carboxytetramethyl Rhodamine | > Dansyl | > EDANS |
| > Cy3/5 | > Mca | > Rhodamine B |
| > Cyanines | > ATTO dyes | > Alexa dyes |
| > Abz-Dnp | > EDANS-Dabcyl | > Mca-Dnp |
| > Tryptophan-Dnp | > FAM-Dabcyl |
The choice of labeling site and conjugation chemistry determines not only labeling efficiency but also the likelihood of preserving peptide bioactivity. Enterprise users typically evaluate these options based on functional risk, site selectivity, and downstream assay requirements. The following table outlines commonly used labeling positions and their professional considerations.
| Labeling Site | Common Reactive Handle | Typical Conjugation Chemistry | Advantages | Key Considerations (Enterprise) |
|---|---|---|---|---|
| N-terminus | Primary amine | NHS ester amide formation | Simple and broadly applicable for many sequences | May affect activity if N-terminus is part of binding/recognition; control of mono-labeling is important |
| C-terminus | Carboxyl group / engineered handle | Amide coupling (via activation) / handle-based coupling | Can preserve N-terminal function for receptor/epitope interactions | Design-dependent; may require peptide engineering to ensure selectivity |
| Lys side chain | Primary amine (ε-NH2) | NHS ester coupling | Flexible option when N-terminus must be preserved | Multiple Lys can cause heterogeneous products; site control often requires sequence design or orthogonal protection |
| Cys side chain | Thiol (-SH) | Maleimide-thiol coupling (or other thiol-selective routes) | Typically enables site-selective labeling when single Cys is used | Thiol oxidation control and maleimide stability considerations; storage and handling affect reproducibility |
| Bioorthogonal handle (Azide/Alkyne) | Azide or alkyne | Click chemistry (CuAAC / SPAAC) | High selectivity with low interference to peptide function | Chosen when strict site control is needed (e.g., multiplex, sensitive binding assays); method selection depends on assay constraints |
Spacer and linker design plays a critical role in minimizing steric interference, reducing fluorescence quenching, and maintaining peptide function. Proper linker selection is often essential for achieving reliable signal performance in binding assays, live-cell imaging, and complex biological matrices.
| Spacer / Linker Type | Typical Effect | When It's Used | Professional Notes |
|---|---|---|---|
| No spacer (direct dye coupling) | Shortest distance; may increase steric effects | Simple probes where binding site is distant from label location | Fastest design but higher risk of activity loss or quenching depending on peptide context |
| Short alkyl spacer (e.g., Ahx) | Improves accessibility; reduces steric hindrance | Binding assays, receptor ligands, antibody-epitope peptides | Common choice to reduce functional disruption without greatly increasing hydrodynamic size |
| PEG spacer (short/medium) | Improves solubility; reduces hydrophobic dye effects | Live-cell imaging, aqueous assay formats, peptides sensitive to aggregation | Often selected to improve handling and reduce non-specific interactions; length chosen case-by-case |
| Long hydrophilic spacer | Maximizes separation between dye and active motif | High-sensitivity binding studies, crowded targets, sterically restricted sites | Useful when label proximity disrupts function; may change PK/behavior in some systems |
| Cleavable linker (application-dependent) | Allows release under defined conditions | Specialized mechanistic studies or workflows requiring conditional signal design | Should be selected based on experimental justification; requires validation of cleavage behavior in intended matrix |
Robust analytical characterization is essential to ensure the consistency, identity, and performance of fluorescence-labeled peptides. Enterprise customers rely on comprehensive QC data to support internal validation, reproducibility across teams, and long-term project continuity. The table below summarizes standard analytical methods and their practical value.
| QC / Analytical Method | What It Confirms | Why Enterprise Customers Care |
|---|---|---|
| RP-HPLC / UPLC | Purity, separation of labeled peptide from free dye and byproducts | Reduces assay interference and improves batch-to-batch comparability in screening and regulated workflows |
| LC-MS | Molecular weight confirmation; evidence of successful conjugation | Supports identity confirmation and internal QA documentation |
| MALDI-TOF (where applicable) | Mass confirmation for certain peptide/dye systems | Useful orthogonal confirmation depending on peptide size and chemistry |
| UV/Vis Quantification | Concentration estimation; dye-to-peptide ratio calculation (method-dependent) | Helps standardize assay dosing and supports reproducibility across sites/teams |
| Fluorescence Spectral Check | Excitation/emission behavior under test conditions | Ensures practical compatibility with instrument filters and detects major quenching issues early |
| Stability / Handling Assessment (as requested) | Behavior under storage/temperature/light considerations relevant to use | Reduces failure in long experiments, multi-week studies, and distributed team workflows |
Application-Focused Design
Labeling strategies are developed based on real assay, imaging, or biological use cases rather than generic dye attachment, improving functional relevance.
Controlled Site-Specific Labeling
Rational selection of labeling sites minimizes disruption of peptide binding, signaling, or uptake behavior.
Broad Fluorophore Compatibility
Support for visible, far-red, and near-infrared dyes compatible with microscopy, flow cytometry, plate readers, and in vivo imaging systems.
Reduced Experimental Variability
Integrated synthesis, labeling, and purification workflows help ensure consistent dye-to-peptide ratios and batch reproducibility.
Flexible Scale Support
From exploratory research batches to larger, consistency-critical production runs for screening or translational studies.
Rigorous Analytical Verification
Each labeled peptide is characterized by appropriate analytical methods to confirm identity, purity, and fluorescence performance.
Scientific Collaboration
Direct access to experienced peptide chemists supports design decisions, troubleshooting, and project evolution.
Documentation & Traceability
Clear analytical reporting supports internal validation, cross-team collaboration, and long-term data reliability.
One-Stop Technical Solution
Integrated peptide synthesis, labeling, purification, and characterization reduce coordination burden and development risk.
Our workflow is designed to balance technical rigor with project efficiency, ensuring that fluorescence-labeled peptides meet functional, analytical, and reproducibility requirements at each stage. The process emphasizes early risk control, clear communication, and traceable outcomes.
1
Project Consultation & Labeling Strategy Definition
2
Peptide Synthesis & Functional Handle Introduction
3
Controlled Fluorescent Labeling
4
Purification & Analytical Characterization
5
Documentation, Delivery & Ongoing Support
Fluorescence and dye-labeled peptides are versatile molecular tools used across life science research, diagnostics, and drug development. By enabling direct visualization, quantitative detection, and functional tracking, labeled peptides support a wide range of experimental workflows where specificity, sensitivity, and reproducibility are critical.
Whether you are developing fluorescence-based assays, imaging probes, or peptide tools for translational research, Creative Peptides provides scientifically grounded fluorescence and dye labeling solutions tailored to your project needs. Our team combines peptide chemistry expertise with application-driven design to support reliable, reproducible outcomes. Contact us today to discuss your requirements, request technical consultation, or obtain a customized quotation.
Fluorescence labeling offers advantages such as high sensitivity, real-time detection capabilities, and compatibility with various imaging techniques like fluorescence microscopy and flow cytometry. It's valuable for studying peptide localization, interactions, and dynamics.
Commonly used fluorophores include fluorescein isothiocyanate (FITC), rhodamine derivatives, cyanine dyes (e.g., Cy3, Cy5), Alexa Fluor dyes, and various other commercially available fluorophores. Selection depends on factors like emission spectrum, photostability, and compatibility with experimental conditions.
Yes, most peptide labeling services offer customization options, allowing you to select the fluorophore and the site of labeling on the peptide molecule based on your experimental requirements.
Fluorescence labeling methods may include amine coupling, thiol-maleimide chemistry, click chemistry, or enzymatic labeling techniques. Each method offers specific advantages and may be chosen based on the peptide sequence and desired labeling site.
Yes, peptides can be labeled with multiple fluorophores either simultaneously or sequentially, allowing for multiplexed detection and imaging in complex biological systems.
Turnaround time varies depending on factors such as the complexity of the labeling process, the number of peptides, and the specific requirements of the customer. However, it generally ranges from a few days to a couple of weeks.
Labeled peptides are typically purified using techniques like HPLC to remove unreacted dye molecules and ensure high purity. Quality control measures often include analytical techniques such as mass spectrometry and spectroscopic analysis to confirm the identity and purity of the labeled peptides.
Storage and handling recommendations may include storing labeled peptides at specific temperatures, protecting them from light exposure, and avoiding multiple freeze-thaw cycles to maintain their stability and fluorescence properties.

References