Innovative Conjugation TechnologyDiverse Coupling StrategiesAdvanced Equipment
Research in modern biomedical and chemical fields now uses peptide molecule coupling technology as a fundamental instrument which creates extensive opportunities for developing new drugs and diagnostic reagents along with biomaterial innovations and other areas. Creative Peptides delivers tailored peptide conjugation solutions to worldwide clients through cutting-edge technology and extensive experience provided by their exceptional team.
Peptides consist of short amino acid sequences that fulfill vital functions in biological systems while also serving as adaptable molecules across different applications. Despite their potential, peptides face practical limitations because they demonstrate poor stability and undergo rapid degradation. Functional group attachment to peptides creates enhanced properties and new applications for these molecules. The peptide conjugation method involves creating covalent bonds between a peptide and other molecules like proteins or drugs and fluorescent probes. A variety of chemical reactions enable this process including amide bond formation thiol-maleimide coupling and click chemistry.
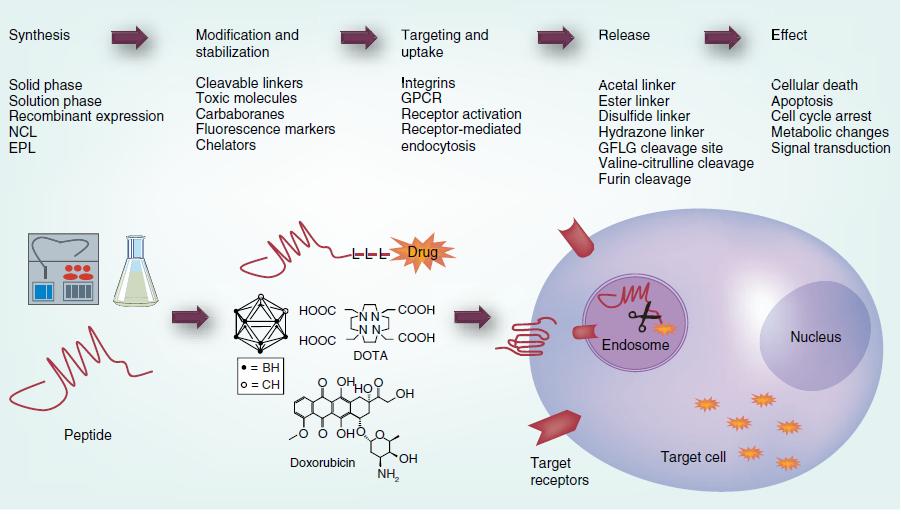 General approach to peptide therapeutics (VM Ahrens, et, al. 2012)
General approach to peptide therapeutics (VM Ahrens, et, al. 2012)
Peptides can be prone to degradation by enzymatic actions and other factors in both in vivo and in vitro environments. Conjugating them with stable molecules significantly improves their stability under physiological or pathological conditions, extending their half-life in the body and enhancing their biological function.
By conjugating peptides with molecules that possess specific targeting abilities, the composite molecules can precisely locate and bind to target cells, tissues, or organs. This improves the therapeutic effect of drugs and reduces side effects on normal tissues. For example, conjugating peptides with antibodies enables specific recognition and targeted therapy of tumor cells.
The combination of peptides with other molecules can produce synergistic effects, granting the composite molecule new biological activity or physicochemical properties. For instance, conjugating peptides with fluorescent molecules allows for cell imaging and biomarker applications; conjugating them with magnetic nanoparticles enables the development of contrast agents for magnetic resonance imaging (MRI).

We utilize a range of high-precision instruments such as state-of-the-art peptide synthesizers and high-performance liquid chromatographs (HPLC) as well as mass spectrometers (MS) and nuclear magnetic resonance spectrometers (NMR). These instruments enable accurate management of peptide coupling processes together with superior product analysis. The peptide synthesizer produces peptide chains that match the predefined sequence and delivers high-quality raw materials for subsequent coupling reactions. Scientists use HPLC and MS to determine product purity and molecular weight throughout the coupling reaction stages while monitoring product progress and quality as it happens. NMR analysis provides detailed insights into the three-dimensional structures of peptide coupling products to advance our knowledge of structure-activity relationships.

We created multiple peptide coupling strategies based on diverse customer requirements and project goals.
Chemical Coupling: The chemical coupling method operates through the application of reagents including carbodiimides and N-hydroxysuccinimide. These chemical reactions between peptides and active functional groups such as carboxyl, amino, or thiol establish stable covalent bonds through catalysis. This coupling method provides benefits including mild reaction conditions and straightforward operation while maintaining broad applicability which allows peptides to efficiently join with proteins, polysaccharides, lipids and various other molecules.
Biological Coupling: The use of biological enzymes like proteases and glycosyltransferases enables precise bond formation between peptides and other biomolecules. We use biological enzymes to achieve the specific coupling of peptides with other biomolecules. Biological coupling achieves high specificity and stereoselectivity which facilitates the production of biologically active molecules with defined structures and functions. Drug development and biopharmaceutical production benefit from this system because it requires high biological activity.
Click Chemistry Coupling: The copper-catalyzed azide-alkyne cycloaddition reaction forms the basis of this method which allows the rapid and effective linkage of peptides with azide or alkyne group-containing molecules. The Click chemistry coupling process utilizes mild reaction conditions to achieve high selectivity while minimizing side reactions to produce stable and biocompatible products. This technique finds extensive application in the fields of biomolecule labeling as well as drug development and materials science.

Our peptide coupling service utilizes a strict quality control system at all production phases which guarantees that the final products achieve our customers' high standards. The entire process from raw material procurement and inspection through peptide synthesis and coupling reactions to purification and separation along with packaging and storage of final products follows strict control and documentation guidelines. Our quality control process uses accepted international standards and testing methods to evaluate important product features including purity and biological activity before delivering customers detailed inspection reports that ensure product quality confidence.
Personalized Customization Services
We gain an in-depth understanding of the client's specific needs and project objectives, providing personalized peptide conjugation solutions, including peptide sequence design, conjugate molecule selection, and reaction condition optimization, to ensure that the final product meets the client's unique requirements.
Comprehensive Technical Support
Our R&D team consists of experts in peptide chemistry, biochemistry, molecular biology, and other fields, with extensive theoretical knowledge and practical experience, allowing us to offer comprehensive technical support and consulting services to clients. We provide one-stop services from project initiation, solution design, experimental implementation to result analysis, helping clients overcome technical challenges in peptide conjugation projects and accelerate the R&D process.
Rapid Delivery and Efficient Service
We have optimized the peptide conjugation service workflow and processes, improving service efficiency and delivery speed, enabling us to complete client projects as quickly as possible while ensuring product quality. This helps clients shorten their R&D cycles.
High Purity and Stability
Through advanced purification technologies and stringent quality control, we ensure that peptide conjugates have high purity, low impurity content, and meet the high standards required for drug development, diagnostic reagent development, and other fields.
Precise Targeting
By utilizing the targeting properties of peptides, conjugating them with other molecules enables precise targeting of specific cells, tissues, or organs, improving the therapeutic effect of drugs and reducing side effects on normal tissues.
Multifunctionality
Peptide conjugates offer multiple functionalities, such as drug delivery, bio-marking, and imaging diagnostics, to meet diverse client needs in different application scenarios. We can functionalize peptide conjugates according to project requirements, such as introducing fluorescent groups, magnetic nanoparticles, and other modifications to provide the product with new functions and characteristics.
Chemical modification of peptide drugs, such as conjugation with polyethylene glycol (PEG), can significantly enhance the stability and bioavailability of the drug, prolong its circulation time in the body, thereby improving its efficacy and reducing the frequency of administration. For example, PEGylated peptide drugs currently on the market have demonstrated good clinical efficacy in treating diseases such as cancer and hepatitis.
The preparation of immunodiagnostic reagents involves conjugating peptide antigens with markers like enzymes or fluorophores to detect disease markers including tumor markers and infectious disease pathogens. This immunodiagnostic method demonstrates high sensitivity and specificity while remaining simple to operate which enables its extensive use in clinical diagnostic practices and disease screening procedures.
Imaging materials such as magnetic nanoparticles or quantum dots combine with peptides to create molecular imaging probes for applications in MRI and PET techniques. Diagnostic probes that target diseased tissues enable early detection and accurate disease localization which helps clinicians make critical treatment decisions.
Researchers attach peptides to biodegradable polymers including poly-lactic acid-glycolic acid copolymers and collagen to form bioactive tissue engineering scaffolds. to create bioactive tissue engineering scaffold materials. The scaffold design replicates the extracellular matrix environment which leads to enhanced cell adhesion and growth and differentiation while speeding up tissue repair and regeneration processes. These materials demonstrate significant potential applications in bone tissue engineering together with cartilage tissue engineering skin tissue engineering and several other areas.
Scientists can build drug sustained-release carrier systems by linking peptides with different materials. When peptides are conjugated with liposomes or nanoparticles it allows for gradual drug release which enhances the duration of drug performance inside the body thereby boosting therapeutic effectiveness and minimizing side effects. Cancer chemotherapy and chronic disease treatment benefit significantly from these drug delivery systems as well as other medical fields.
Engage in in-depth discussions with the client to understand their specific needs, including the peptide sequence, length, purity requirements, the object to be conjugated (such as proteins, small molecule drugs, carbohydrates, etc.), conjugation methods, and application scenarios.
Based on the client's needs, design a peptide conjugation plan, including peptide synthesis routes, conjugation reaction conditions, purification methods, etc., and discuss and confirm the plan with the client.
According to customer needs, different methods are used to complete the synthesis of peptides.
Use different modification methods according to actual conditions. (N-terminal Modification, C-terminal Modification and Side-chain Modification)
Adopting diverse coupling strategies.
Purity Testing, Molecular Weight Determination, Amino Acid Sequence Analysis and so on.
Deliver the peptide conjugation products along with related quality testing reports (such as HPLC chromatograms, MS data, etc.) to the client.
Peptide-drug conjugates (PDCs) are metabolized and excreted through several pathways in the body. After binding to receptors on the target cell surface, PDCs undergo target-mediated drug elimination, where they are internalized and degraded in lysosomes. Additionally, PDCs can be released through nonspecific metabolism via protease hydrolysis or nonspecific endocytosis. Due to their smaller molecular weight, PDCs may also be rapidly cleared by the kidneys through renal excretion.
Peptide-drug conjugates generally have low immunogenicity, but they may still trigger the production of anti-drug antibodies (ADA), leading to drug clearance by the immune system.
Commonly used carrier proteins include KLH (keyhole limpet hemocyanin), BSA (bovine serum albumin), and OVA (ovalbumin). Among them, KLH is widely used due to its high antigenicity.
Cell-penetrating peptides (CPPs) are peptides that enable oligonucleotides to enter cells through several mechanisms. They first bind to the negatively charged components of the cell membrane via electrostatic interactions. CPPs can also induce direct membrane penetration by forming temporary pores, allowing oligonucleotides to pass through the membrane. Additionally, CPPs can facilitate endocytosis by forming complexes with oligonucleotides, which are then internalized by the cell. Once inside, CPPs can disrupt the lysosomal membrane, enabling the release of oligonucleotides into the cytoplasm, where they can continue their function.
