To improve peptide pharmacokinetics, conjugating the peptide to lipids is a favored approach. Peptides-Fatty acids conjugation is obtained by fatty acid modification of the main chain structure or contralateral chain group of polypeptide drugs. Creative Peptides specializes in providing high-quality and reliable peptides-fatty acids conjugation services to support various research and development projects.
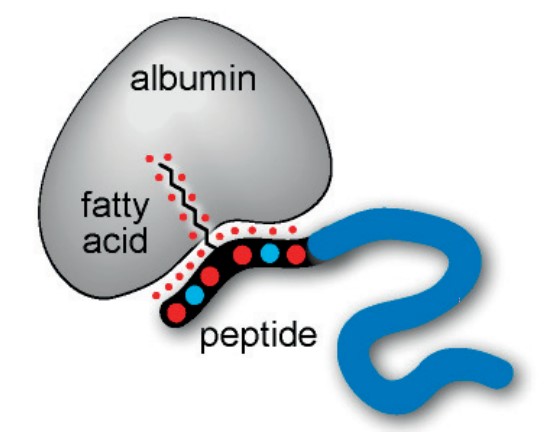 Fig. 1. Schematic representation of a bioactive peptide (in blue) modified with a peptide-fatty acid chimera. Interactions involved in the binding from both fatty acid and peptide are shown as red dots. (Zorzi, A., 2017)
Fig. 1. Schematic representation of a bioactive peptide (in blue) modified with a peptide-fatty acid chimera. Interactions involved in the binding from both fatty acid and peptide are shown as red dots. (Zorzi, A., 2017)
Type of fatty acid includes: Caprylic acid (C8), Capric acid (C10), Lauric acid (C12), Myristic acid (C14), Palmitic acid (C16) or Stearic acid (C18) etc. Peptides-fatty acids conjugation drugs can improve the liposolubility, intestinal mucosal permeability and absorption efficiency. It prolongs the half-life in the circulation significantly. Fatty acid conjugated peptides can also be used for a number of different applications, for example, Detemir (Levemir) is a new insulin analogue developed by Northrop. After the removal of human insulin B30 Thr, a myristic acid side chain was attached to B29 Lys, which could release the drug slowly and achieve the effect of long-term hypoglycemia.
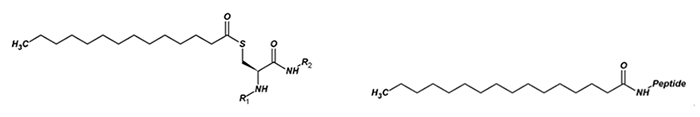 Fig. 2 The structure of myristoyl thioester and palmitoyl ester.
Fig. 2 The structure of myristoyl thioester and palmitoyl ester.
Peptides and fatty acids can be conjugated together through various chemical techniques to increase the functionality and efficiency of the peptides in biomedical applications. Some of these methods include:
Direct Conjugation: Conjugation can occur via two primary methods - amide linkage and ester linkage. In amide linkage, the carboxyl group of the fatty acid reacts with the amino group of the peptide. The reaction between these two groups forms an amide bond, a stable bond that can resist breakdown in the body. On the other hand, in ester linkage, the carboxyl group of the fatty acid reacts with the hydroxyl group of an amino acid side chain in the peptide. This reaction forms an ester bond. Ester bonds are less stable and can be hydrolyzed in the body by enzymes called esterases. The choice of linkage may depend on the intended application. For example, if a slow-release effect is desired, an ester linkage could allow for gradual breakdown and release of the peptide. If resistance to breakdown is desired, for instance, for systemic delivery of the peptide, an amide linkage might be more appropriate.
Amide Bond Formation using Coupling Agents: This is another method where in, coupling agents like carbodiimide (EDC) or N-hydroxysuccinimide (NHS) are used to activate the carboxyl group of the fatty acid. Then, the activated carboxyl group reacts with the amine group of the peptide to form an amide bond.
Thioether Linkage: This method involves conjugating the fatty acid to the peptide using a cysteine amino acid which can form a thioether bond with the fatty acid.
Click Chemistry: In this method, azide group attached to fatty acid and alkyne group attached to peptide react with each other under copper(I) catalysis to form a stable triazole linkage.
Liposome Fusion: In this technique, liposomes are loaded with a fatty acid and fused with a peptide. The peptide and fatty acid then conjugate to form a peptide-fatty acid complex.
Solid Phase Peptide Synthesis (SPPS): Fatty acids can be conjugated to a peptide via solid phase peptide synthesis. This technique allows the addition of specific fatty acids to certain amino acids in the peptide chain.
No matter what method is used, the conjugation of fatty acids to peptides needs to be performed in a controlled manner to ensure specificity, and the result product always needs to be purified and verified through analytical methods, such as mass spectrometry or nuclear magnetic resonance (NMR) spectroscopy.
Improve Biological Activity and Stability: By combining fatty acids with peptides, the biological activity and stability of peptides can be increased. Fatty acids can protect peptides from enzymatic hydrolysis and other biodegradation processes, thus prolonging the half-life of peptides in vivo and improving the therapeutic effect. Fatty acid conjugation of GLP-1 analogues has been shown to increase their half-life from 10 to 12 hours to days
Improve Solubility and Bioavailability: Some peptides may have poor solubility in water due to their hydrophobicity or polarity. Combined with fatty acids, it can improve the solubility of peptides, improve their bioavailability in vivo, and make them easier to be absorbed and utilized.
Enhance Plasma Albumin Binding Rate: Fatty acid can reversibly bind to human serumalbumin (HSA) in vivo. The combined complex is restricted by excessive molecular size in transmembrane transport. Therefore, fatty acid modification can prolong the retention time of drugs in vivo.
Enhance Targeting and Permeability: Fatty acids usually have specific targeting and permeability and can guide peptides to specific tissues or cells. This targeted delivery can reduce the side effects of drugs on non-target tissues and improve the therapeutic effect. At the same time, fatty acids can also promote peptides to penetrate the cell membrane and enter the cell interior, playing a therapeutic role.
Multifunctional and Synergistic Effect: Peptide-fatty acid conjugate can have multiple biological activities of peptide and fatty acid, and realize the synergistic effect of multiple functions. This synergistic effect can further enhance its therapeutic effect and meet the treatment needs of complex diseases.
Flexibility and Customizability: The preparation process of peptide-fatty acid conjugate is relatively flexible, and different peptides and fatty acids can be selected for combination according to specific needs. This customization enables the peptide-fatty acid conjugate to be personalized for specific diseases or physiological processes, and improves the therapeutic effect and safety.
Drug Delivery: One of the most significant applications of peptides-fatty acids conjugation is in the field of drug delivery. By combining a fatty acid with a peptide, a compound that is more easily absorbed by the body is created. It can also be used as a targeted drug delivery system to accurately deliver drugs to the lesion site, improve the therapeutic effect and reduce side effects. This can help to improve the efficacy of various drugs, making them more effective in treating a wide range of conditions.
Vaccine Design: Peptides conjugated with fatty acids have been used to enhance immune responses in vaccine design. They have been shown to induce stronger and longer-lasting immune responses.
Treatment of Cancer: Certain peptides have anti-cancer properties and their conjugation with fatty acids can further enhance their potency. The conjugates can selectively target and kill cancer cells, while leaving healthy cells unharmed.
Treatment of Neurodegenerative Diseases: Peptides-fatty acids conjugates are being explored for the treatment of neurodegenerative diseases such as Alzheimer's. Research has shown that these conjugates can pass the blood-brain barrier, enabling them to deliver drugs directly to the brain.
Detection of Diseases: Peptides-fatty acids conjugation can also be used in diagnostic techniques for the detection of various diseases, including cancer and infectious diseases. The conjugates can act as probes to detect specific biomarkers associated with these diseases.
Antimicrobial Agents: Peptides-fatty acids conjugates have been used as effective antimicrobial agents. They can interact with the bacterial cell membrane, leading to its destabilization and the death of the bacteria.
Pep19-4LF is a synthetic antibacterial peptide with high antibacterial activity. However, the key motif of this peptide shows some limitations, such as poor solubility, low synthetic yield and aggregation in biological media. Chen et al. solved these limitations by replacing problematic motifs with fatty acids. The modified peptide showed enhanced antibacterial activity, and the length of fatty acid chain was directly related to this activity. It shows high antibacterial activity and no toxicity, and shows the prospect as a potential drug after being proved successful in early rodent experiments. It is concluded that replacing the hydrophobic motif of peptide with a medium-length fatty acid simplifies its design and maintains high antibacterial activity.
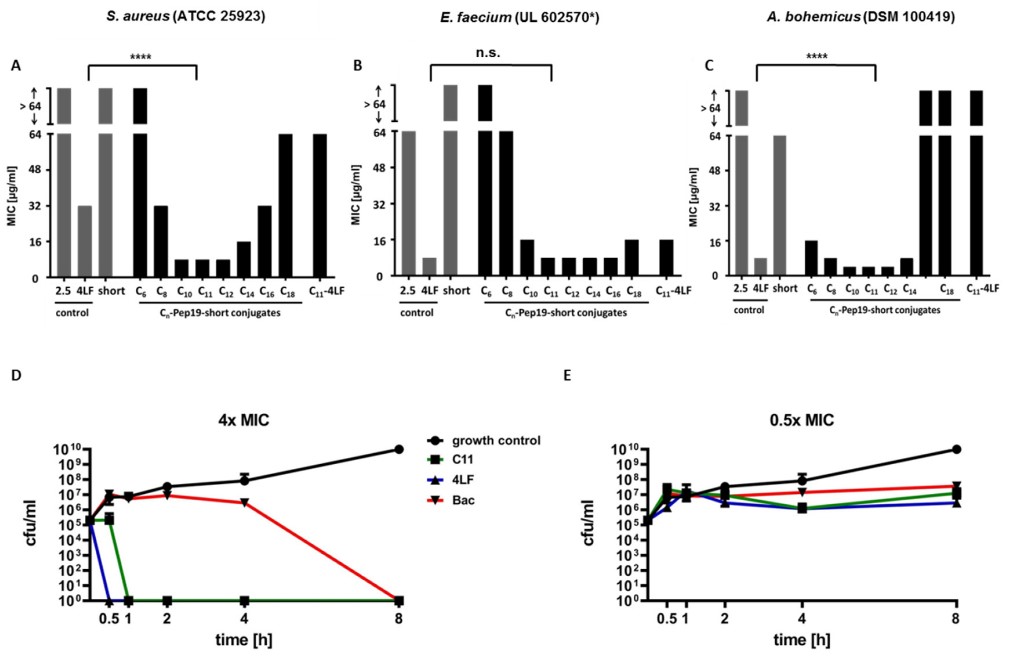 Fig. 3 MIC values of Pep19-short conjugated to different fatty acids and time-kill studies of the most potent fatty acid conjugate (1-Pep19-short) on different bacteria. (Storck, P., 2020)
Fig. 3 MIC values of Pep19-short conjugated to different fatty acids and time-kill studies of the most potent fatty acid conjugate (1-Pep19-short) on different bacteria. (Storck, P., 2020)
Nutrition and Health Supplements: Peptides-fatty acids conjugates can be used in functional foods, nutrition, and health supplements for their potential health benefits, including anti-inflammatory, anti-oxidant, and anti-ageing properties.
Cosmetic and Personal Care Products: Due to their skin penetration enhancing properties, peptides-fatty acids conjugates are often used in cosmetic and personal care products to deliver active ingredients deeper into the skin.
Creative Peptides offers comprehensive peptides-fatty acids conjugation services, including but not limited to:
Custom Peptide Synthesis: We provide a reliable and efficient service for the custom synthesis of peptides, tailored to your research needs.
Conjugation Services: Our team is proficient in the preparation of fatty acid-peptide conjugates, implementing advanced conjugation techniques to ensure the quality and consistency of our services.
Analytical Services: We ensure the accuracy and precision of our peptides-fatty acids conjugation through specialized analytical services. These include Mass Spectrometry, High-performance Liquid Chromatography (HPLC), and more.
Stability Studies: We also conduct stability studies to assess the stability of the peptide-fatty acid conjugates under various conditions.
Scale-Up Production: Our well-equipped facility can handle multi-stage scale-up production starting from milligrams to grams, ensuring continuity for your research or product development.
Documentation and Support: We further offer thorough documentation of our synthesis and conjugation procedures, as well as comprehensive support throughout the entire project to ensure maximum customer satisfaction.
Consultancy Services: With our team of experts, we are able to offer advice and consultancy on the most efficient and effective methods for your specific peptides-fatty acids conjugation needs and requirements.
Our team utilizes advanced chemistry and state-of-the-art technology to deliver the highest quality peptides-fatty acids conjugation services, catering to various research, medical, and pharmaceutical industries.
The conjugation of peptides with fatty acids enhances their stability, solubility, and cellular uptake. It can improve peptides' potential as therapeutics by enabling them to penetrate cells more effectively and resist rapid degradation. It also helps to facilitate targeted delivery of peptides and enhance their interaction with cell membranes.
We adhere to rigorous protocols and standards during our conjugation process. Our team of skilled scientists conducts all processes in a highly controlled environment, and each step's end products are thoroughly verified for quality. All conjugates undergo rigorous QC testing, including Mass Spectrometry and HPLC report, to ensure purity and correct conjugation.
Peptides-Fatty Acids Conjugates can play a vital role in drug development, mainly when used as therapeutics. Their improved stability and cell penetration abilities can make them effective in delivering therapeutic agents directly to the requisite cells. Moreover, their enhanced solubility can overcome the challenges related to the bioavailability of therapeutic peptides.
Indeed, we can perform the conjugation with a variety of fatty acids and peptides. We offer a wide range of customization options to cater to different therapeutic needs and research requirements.

References