Backbone cyclization, also known as head-to-tail cyclization, refers to the formation of covalent bonds between the N-terminal and the C-terminal of a peptide. Backbone cyclization is a technology which is used to make backbone cyclized peptides (BCPs). Head-to-tail backbone (homodetic) cyclization represents a simple yet powerful peptide modification that imparts rigidified structure, biorelevant turn conformations, increased proteolytic stability, and improved membrane permeability. The abnormal stability and high biological activity of BCP have made it a hot spot in the field of drug discovery and cyclization technology. Creative Peptides is committed to providing high-quality, high-precision backbone cyclization services. Our service allows researchers to take advantage of the characteristics of head-to-tail cyclized peptides and apply them to the improvement of existing drugs or the development of new drugs.
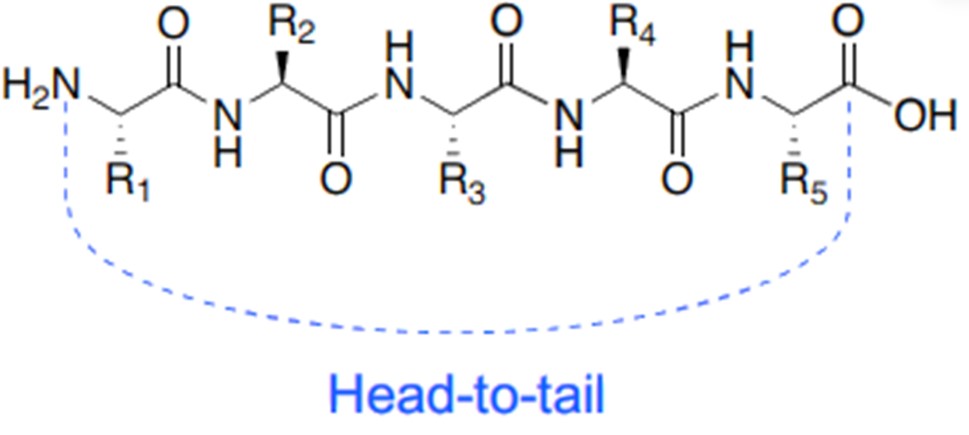 Fig.1 Head-to-tail cyclization. (Buckton, L. K., 2021)
Fig.1 Head-to-tail cyclization. (Buckton, L. K., 2021)
Backbone-cyclized peptides are easily assembled via the formation of an amide bond between the peptides own N-terminus amine and C-terminus carboxylic acid. There are three ways to obtain backbone cyclized peptides: isolation from organism, gene recombination technology and chemical de novo synthesis. The method of purifying backbone cyclized peptides from organism has the disadvantages of high separation difficulty, low efficiency and heavy workload. Because backbone cyclized peptide has a special head-to-tail cyclic amide bond structure, the traditional gene recombination expression technology can not be directly used for the preparation of backbone cyclized peptide, so the strain needs to be rebuilt. Therefore, the technical difficulty of this method is relatively high. The toxicity of backbone cyclized peptide to strains often leads to the failure of gene recombinant expression. In addition, the ability of biological expression of backbone cyclized peptide containing unnatural modification is very limited. Compared with the first two methods, chemical de novo synthesis of backbone cyclized peptide has advantages, such as arbitrary change of amino acid sequence, arbitrary mutation of site and arbitrary insertion of unnatural amino acids.
Solid-phase cyclization means that the end-to-end amide bond reaction of polypeptide is completed on resin. This strategy has two obvious advantages, such as reducing the probability of intermolecular collision and reducing the side reaction of intermolecular polymerization; Generally speaking, peptides can be immobilized on the resin in two ways, namely, anchoring the resin with side-chain functional groups and immobilizing the resin with C-terminal carboxyl groups. After splicing the polypeptide sequence on the solid phase, the former removes the N-terminal and C-terminal protective groups to release free amino groups and carboxyl groups. Then, the condensation cyclization of C-terminal carboxyl group and N-terminal amino group is completed on the solid phase. The latter uses the carboxyl group of the main chain to anchor the resin. After the splicing of the polypeptide sequence is completed, the C-terminal of the polypeptide is activated, and then the intramolecular amine cyclization reaction occurs with the N-terminal free amino group. The second method is carried out simultaneously with the solid phase cleavage of the polypeptide, so it is superior to the first method. Solid-state synthesis includes side chain fixed resin, sulfonamide-terminated fixed resin, aryl acyl hydrazine-terminated fixed resin and other methods.
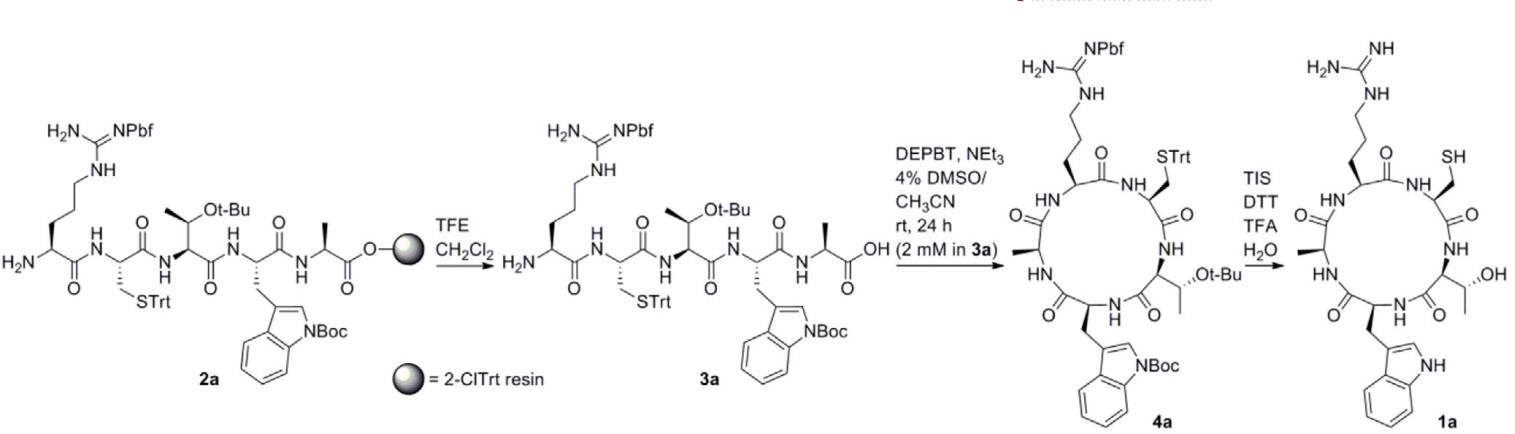 Fig. 2 Resin used in synthesis of head-to-tail cyclic peptide 1a. (Bouayad-Gervais, S., 2022)
Fig. 2 Resin used in synthesis of head-to-tail cyclic peptide 1a. (Bouayad-Gervais, S., 2022)
Solid-phase cyclization strategy is not suitable for the synthesis of BCPs with long chain. This is because the efficiency of solid-phase reaction will be significantly reduced with the growth of peptide chain. Therefore, the efficient synthesis method for long-chain BCPs is still liquid-phase condensation. The traditional liquid-phase condensation takes linear peptides with fully protected side chains as raw materials and completes the head-to-tail cyclization of peptides through condensation reaction. The solubility of fully protected side chains is greatly influenced by sequence and chain length. Therefore, it is necessary to optimize the organic solvent before condensation. In order to reduce the side reaction of intermolecular condensation, the concentration of liquid phase condensation is generally required to be strictly controlled. Moreover, the traditional condensation reaction sites are generally suitable for backbone cyclization reactions with Gly and Pro at the C-terminal. With the development of peptide chain docking technology, chemical selective reaction has become a new method for the synthesis of BCPs. Compared with traditional condensation reactions, chemical selective reaction has higher stereoscopicity and more mild and efficient reaction conditions, so it has become the mainstream of recent research in this field.
In 1994, the natural chemical linking reaction developed by Kent et al. was the most widely used chemical synthesis method of protein. That is, in the presence of pH 7 and mercaptan, a peptide chain with thioester at the C-terminal and another peptide with cysteine at the N-terminal could have an efficient amide bond linking reaction. The natural chemical linking reaction was completed through two steps: chemical selective capture and intramolecular rearrangement. First, Reversible mercaptan-thioester exchange takes place between C-terminal thioester and N-terminal Cys side chain sulfhydryl group of two molecules to generate a thioester intermediate of one molecule, and then, natural peptide bond is generated through irreversible S-N acyl migration in the molecule. The advantages of natural chemical connection reaction are: high efficiency, high selectivity, and no need to protect the side chain. BCPs can be prepared by natural chemical connection reaction in the molecule. That is, a linear peptide containing both C-terminal thioester and N-terminal Cys can be connected from head to tail through intramolecular amide bond reaction. In 1997, Tam et al. synthesized plant cyclic polypeptide Cyclotide for the first time by intramolecular natural chemical connection reaction. Subsequently, many researchers completed the synthesis of backbone cyclic peptide by natural chemical connection reaction. In principle, natural chemical connection cyclization strategy belongs to one of liquid phase cyclization reactions.
The natural function of the BCPs is host defense, that is, to protect the survival and continuation of this species by inhibiting the growth of alien organisms. Through the activity experiments, the researchers found that the BCPs also have some unique biological functions, which could be applied in treating human diseases. For example, the cyclotide in plants has the properties of uterine contractile activity, antimicrobial activity, anti-HIV activity and anti-tumor activity. However, the BCPs derived from organisms may be biotoxic to humans. In order to reduce the biotoxicity, the researchers will either mutate individual sites or fuse biologically active exogenous peptides into BCPs by "grafting".
Custom synthesis: We offer the synthesis of backbone cyclized peptides as per your specifications in desired purity and quantity levels. The following is our classic synthesis case.
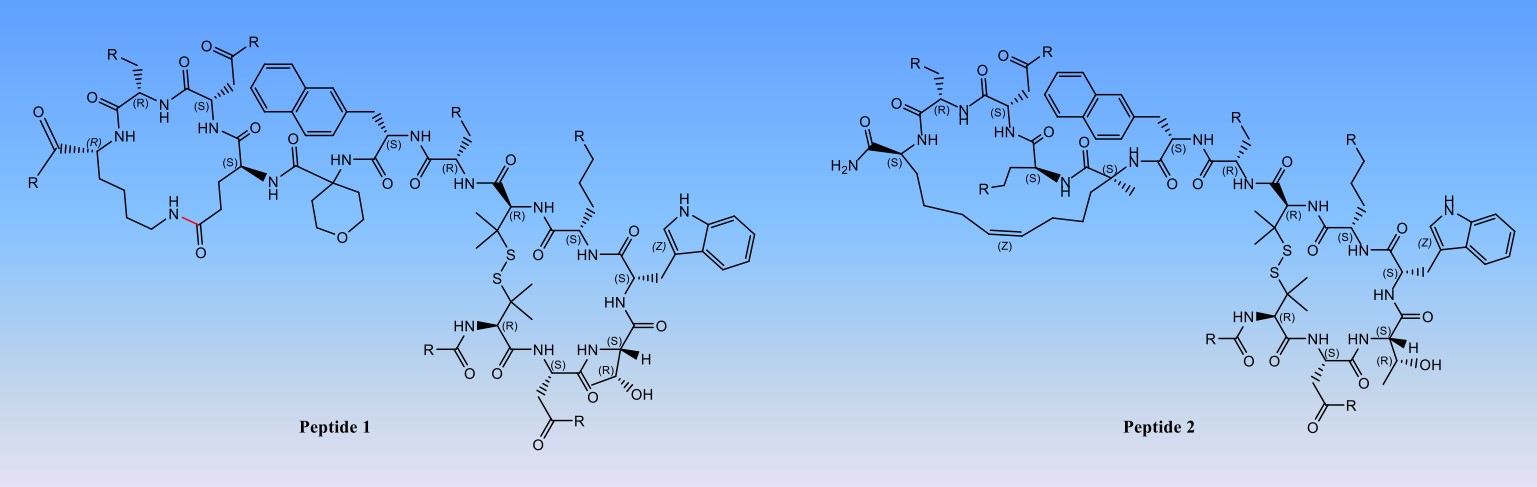 Fig. 3 Synthesis of peptide molecules with head to tail rings.
Fig. 3 Synthesis of peptide molecules with head to tail rings.
Peptide design and optimization: We provide peptide design and solutions to improve the stability, selectivity, potency and production yield of the backbone cyclized peptides.
Peptide modification: We offer various types of peptide modifications such as N-terminal acetylation, C-terminal amidation, phosphorylation, methylation and more to enhance the properties of cyclized peptides.
Peptide scale-up production: Our production facilities can scale up the synthesis of backbone cyclized peptides to meet the needs of our clients for preclinical and clinical studies.
Analytical services: We offer comprehensive analytical services including high performance liquid chromatography (HPLC), mass spectrometry (MS), amino acid analysis, purity checks (QC) and more to ensure the quality of synthesized peptides.
Technical support: Our team of expert scientists is available to provide all necessary technical support and consultation on best practices in terms of storage, handling, and use of backbone cyclized peptides. peptides.
Creative Peptides commonly used methods are chemical synthesis and biosynthesis. We perform the reaction with very high yields, and routinely provide over 98% purified cycled peptides. Our experienced scientists will tailor the most appropriate methods and 100% guaranteed service for customers. Every step of peptide synthesis is subject to Creative Peptides' stringent quality control. Typical delivery specifications include:
The typical turnaround time varies depending on the complexity of the peptide sequence, quantity, and the degree of purity requested. Generally, it can take anywhere from 2 to 6 weeks.
Yes, we guarantee the peptide purity which is defined by High Performance Liquid Chromatography (HPLC) analysis. We provide the HPLC report and Mass spectrometry report as well.
All of our peptides are subjected to stringent quality control including HPLC purity testing and accurate mass determination by mass spectrometry.
Yes, we can provide large-scale synthesis of peptides ranging from milligram to kilogram quantities.
Yes, please contact us with your peptide sequence and desired quantity and purity. We will provide a detailed quote based on your requirements.

References