Stapled peptides are a mind-boggling class of peptides that have been painstakingly modified via chemical sorcery to introduce a covalent bond between two amino acids, usually through the use of a hydrocarbon linker that is at once slippery and strong. This creates a staple-like structure that works like a mystical spell to stabilize the peptide in a helical conformation, adding a touch of surrealism to an already intricate biochemical dance. In doing so, the peptide's binding affinity and selectivity for its target protein becomes almost supernatural in its efficacy.
It is worth noting that the staple itself is a small, hydrocarbon-based molecule that is cross-linked between two amino acids in the peptide sequence, and it is this occult cross-linking that effectively locks the peptide into a specific conformation, like a forbidden ritual that traps the peptide in a predestined fate. The unique structure of the stapled peptide can then be optimized to achieve a perfect binding harmony with a specific target protein, resulting in an otherworldly level of binding selectivity and potency that defies the laws of nature.
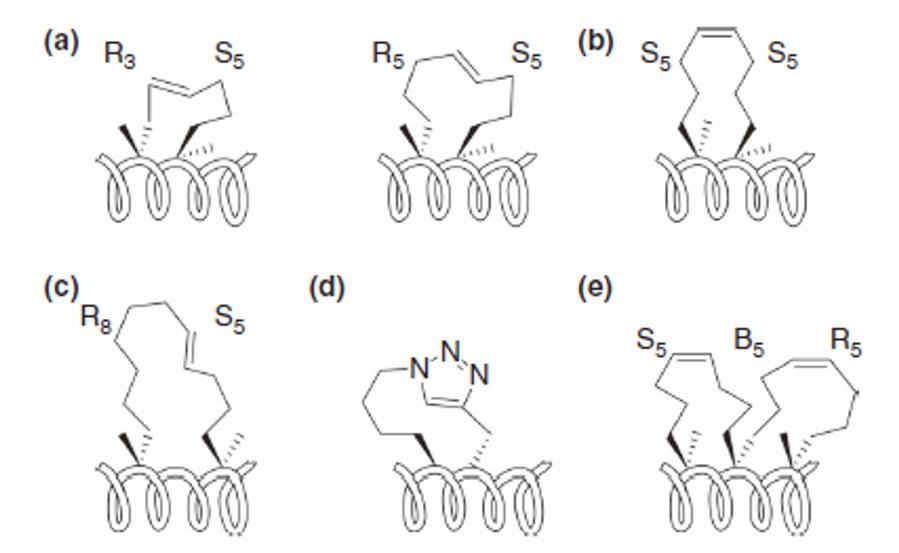 Stapled peptide architectures (Tan Y S, et al. 2016)
Stapled peptide architectures (Tan Y S, et al. 2016)
This modification improves their ability to bind to protein targets and has led to their use in a variety of applications in both basic research and drug development.
Target validation: Stapled peptides are designed to bind to one of the interacting partners, which can serve as an effective tool for validating the biological relevance of a particular protein-protein interaction. By selectively disrupting the interaction, these peptides can help elucidate the functional roles of specific proteins in cellular processes and disease pathways.
Therapeutic development: Stapled peptides are designed to target specific protein-protein interactions or signaling pathways that are dysregulated in the following diseases, including cancer, infectious diseases, and metabolic disorders.
Chemical biology: Owing to their remarkable ability to selectively modulate the activity of target proteins, stapled peptides represent a potent tool for probing the intricate and multifaceted functions of specific protein targets in cells and tissues. By selectively modulating the activity of a target protein, researchers can gain insights into its role in various biological processes.
Imaging: Stapled peptides, labeled with fluorescent or radioactive tags, enable visualization of specific proteins in cells or tissues. This is a good method for studying protein localization and dynamics, as well as for identifying new drug targets.
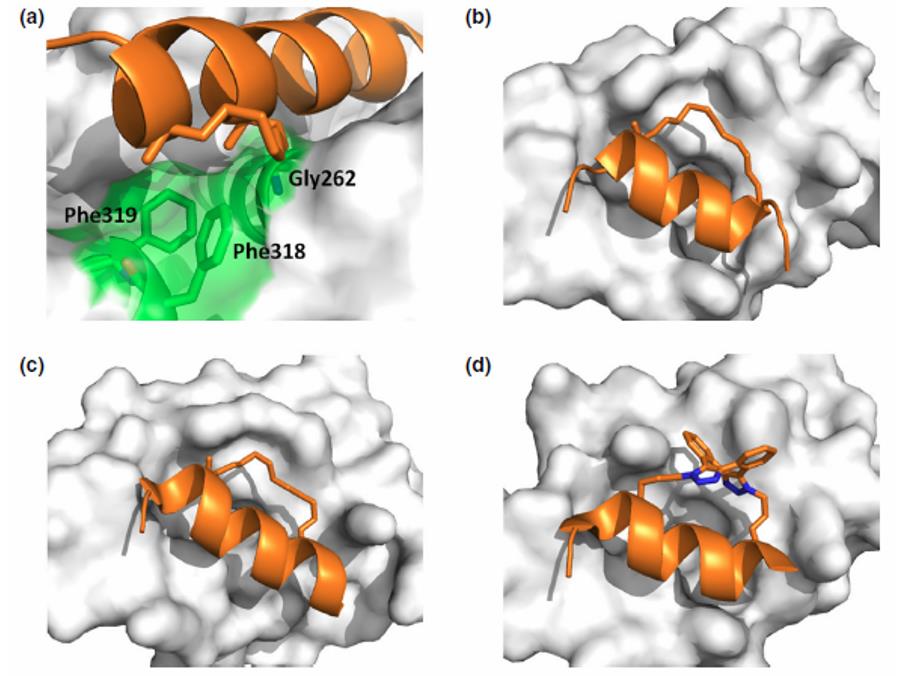 Staple binding sites on target proteins (white) (Tan Y S, et al. 2016)
Staple binding sites on target proteins (white) (Tan Y S, et al. 2016)
The process of stapling can be achieved using a variety of methods, including ring-closing metathesis (RCM), olefin cross-metathesis (OCM), and hydrogenation. RCM is the most commonly used method, as it is efficient and compatible with a wide range of peptide sequences.
The synthesis of stapled peptides is different from that of a common peptide. By use of solid phase methods, the peptide chain is introduced two unnatural amino acids containing an α-methyl group and an α-alkenyl group, and then an olefin metathesis reaction occurs between two unnatural amino acids to constitutes a stable α-helical conformation.
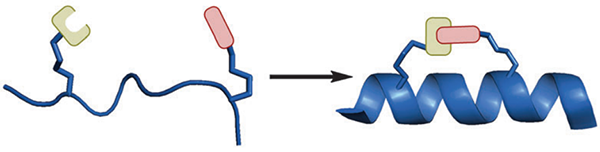
One-component stapling techniques: Using non-native amino acids bearing complimentary side-chain functional groups that can be directly coupled together.
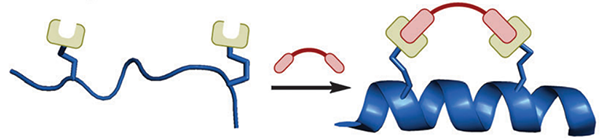
Two-component stapling techniques: Involving a bifunctional linker compound which forms a staple by reacting with two complementary non-native amino acids in the peptide of interest.
Characterizing stapled peptides is critical for assessing their potency, stability, and pharmacokinetic properties. Here are some common methods we used:
Creative Peptides provides efficient and high quality peptide synthesis services while offering different modification services.
Stapled peptides are peptides modified with a covalent bond between two amino acids, typically through a hydrocarbon linker. This modification stabilizes the peptide in a helical conformation, enhancing its ability to bind specifically to target proteins, making them more potent and stable in biological systems.
Stapled peptides are used in various applications, such as target validation by disrupting protein-protein interactions, chemical biology for probing protein functions, and imaging to study protein localization and dynamics. They are also used to gain insights into disease mechanisms and to identify potential drug targets.
Stapling enhances a peptide's stability by increasing its resistance to proteolytic degradation and improving its serum half-life. This modification also boosts cell permeability, allowing peptides to effectively target both extracellular and intracellular proteins with higher binding affinity.
Stapled peptides are synthesized using techniques such as ring-closing metathesis (RCM), olefin cross-metathesis (OCM), and hydrogenation. RCM is the most commonly used due to its efficiency and compatibility with various peptide sequences, enabling precise staple formation.
Designing stapled peptides involves selecting the appropriate staple position, stereochemistry, and length, and making sequence modifications to enhance target binding. Depending on the application, single or multiple staples can be incorporated to optimize peptide functionality and stability.
Stapled peptides are characterized using techniques like mass spectrometry, circular dichroism (CD), nuclear magnetic resonance (NMR), thermal denaturation, and cell-based assays. These methods help assess the peptide's stability, structure, and effectiveness in binding to its target.
