Creative Peptides offers a comprehensive Structure-Activity Relationship (SAR) analysis service covering Quantitative Structure-Activity/Property Relationship (QSAR/QSPR) and 3D-QSAR model construction. SAR analysis aims to reveal the qualitative or quantitative relationship between the molecular structure of a chemical substance and its biological activity or physicochemical properties. Through SAR studies, scientists are able to identify key structural features and their effects on biological activity, providing a scientific basis for drug design and optimization. These methods not only accelerate the development of new drugs, but also improve the efficacy, selectivity, and safety of drugs, and facilitate the innovative development of peptide drugs.
Structure-activity relationship studies the relationship between a molecule's structure and its biological activity or physicochemical properties. SAR can be divided into two forms: qualitative and quantitative. Qualitative SAR infers activity differences through the observation of structural characteristics, while quantitative SAR uses mathematical models to quantify this relationship to predict the potential activity of new compounds. By combining molecular descriptors (e.g., molecular weight, polarity, etc.) with known bioactivity data, QSAR models can help scientists understand the mechanism of compound and target interactions, and play an important role in drug design and optimization. In addition, SAR methods are widely used in environmental science and toxicology studies to predict the health and environmental effects of chemical substances.
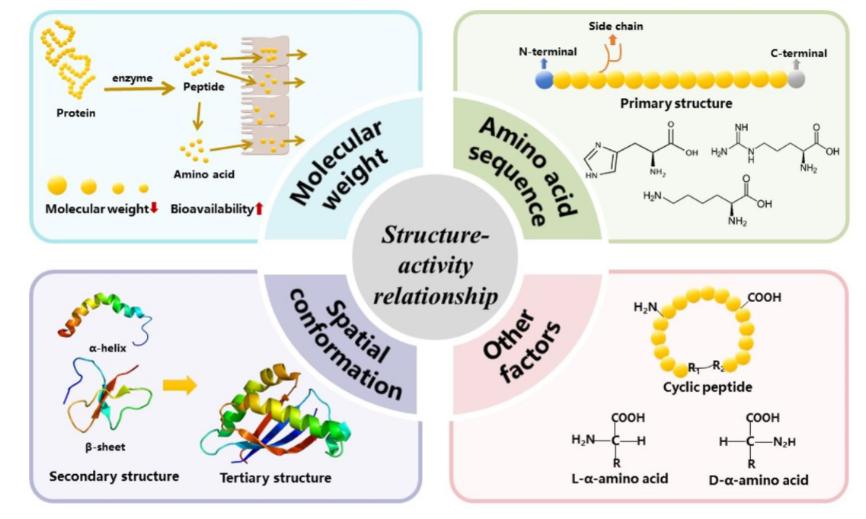 Fig.1 Relationships between structure and activity of bioactive peptides. (Tang, Cai-die, et al., 2024)
Fig.1 Relationships between structure and activity of bioactive peptides. (Tang, Cai-die, et al., 2024)
The earliest study of structure-activity relationship speculated the relationship between the structure and activity of physiologically active substances, and then inferred the structure of the active site of the target enzyme and designed a new structure of the active substance. With the development of information technology, at present, the quantitative structure-activity relationship with the computer as an auxiliary tool has become the main direction of structure-activity relationship research. The QSAR has also become one of the important methods for rational drug design.
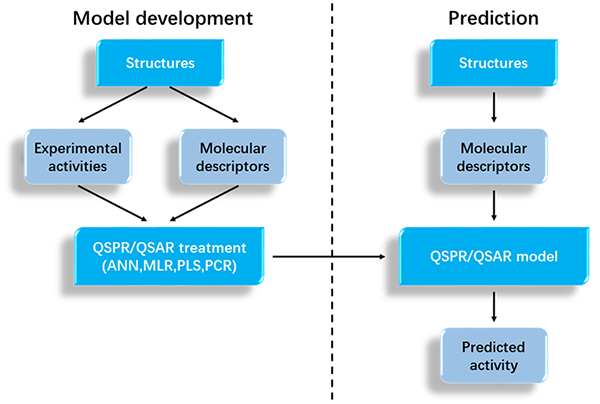 Fig.2 Overview of QSAR/QSPR modeling. (Patel, et al., 2014)
Fig.2 Overview of QSAR/QSPR modeling. (Patel, et al., 2014)
Quantitative Structure-Activity/Property Relationship is the application of chemical theory calculation methods and various mathematical statistical analysis methods to quantitatively describe and study the relationship between the structure and activity/property of organic objects, and establish a mathematical model of QSAR/QSPR. Once the reliable QSAR/QSPR model is established, it can be used to predict the various properties of the new compound after testing for predictive power. The basic assumption in the QSAR/QSPR study is that the characters or biological activity of a compound can be expressed as a function of the chemical structure. That is, a mathematical model can be used to combine theoretical calculations and statistical analysis to study the quantitative relationship between the structure of a series of compounds and their properties.
Peptides have gained renewed interest as candidate therapeutics. However, to bring them to a broader clinical use, challenges such as the rational optimization of their pharmacological properties remain.
In peptide drug discovery, structure-activity relationship studies are crucial. Here are a few real-world R&D examples that demonstrate how peptide drug design and development can be optimized through SAR studies.
Case 1:Chemical modification and activity optimization of Gomesin
Gomesin is a cationic antimicrobial peptide isolated from the blood cells of the Brazilian spider Acanthoscurria gomesiana and exhibits broad toxicity to gram-positive bacteria, gram-negative bacteria, fungi, cancer cells, and parasites. Despite its significant biological activity, its natural form is less stable in human serum, limiting its potential clinical application. Through the study of the structure-activity relationship, scientists have developed a cyclic version of Gomesin, which significantly improves its stability and makes it more resistant to degradation in human serum. In addition, cyclic Gomesin exhibits a stronger ability to penetrate cells and can directly enter cancer cells for targeting. Further chemical modification optimized its molecular structure and improved its antibacterial and anticancer activities. These improvements make Gomesin a novel drug candidate with both potential anticancer and antimicrobial activity, demonstrating the important role of SAR analysis in optimizing drug stability and potency.
Case 2: Structural optimization and application expansion of wasp peptides
Wasp peptide is a short-chain, amphiphilic, α-helix antimicrobial peptide with a variety of biological activities such as antibacterial, antitumor, antifungal and antimalarial. However, it has strong hemolysis and toxicity to normal cells, which limits its medicinal development. Through SAR analysis, scientists employed a variety of structural optimization strategies, including point mutagenesis, truncation of peptide chains, introduction of accessory amino acids at the N-terminus, and replacement of L-amino acids with D-amino acids. These modifications not only significantly improved the biological activity and targeting of wasp peptides, but also reduced the hemolysis to normal cells and improved its safety. The optimized wasp peptide shows greater application potential in the field of antimicrobial and anti-tumor applications. This case shows that SAR studies can optimize drug properties through precise structural modification, thereby expanding their clinical applicability.
Case 3:Design of DPP-IV inhibitors and treatment of diabetes
SAR analysis has played a central role in the development of dipeptidyl peptidase IV (DPP-IV) inhibitors. Through the quantitative structure-activity relationship model, scientists are able to quantify the relationship between molecular structure and inhibitory activity, allowing for the prediction and design of novel peptide inhibitors with high specificity. Specifically, using stoichiometric models and known activity data, the researchers identified peptide sequences with significant inhibitory activity and guided the synthesis and optimization of new compounds. This QSAR-based design approach significantly improves R&D efficiency and reduces the workload of experimental screening. At present, this strategy has been widely used in the development of diabetes treatment drugs, providing a scientific basis for the discovery of more efficient and safer DPP-IV inhibitors, and demonstrating the great potential of SAR analysis in modern drug development.
Creative Peptides has developed a reliable procedure for SAR analysis of peptides, we can provide global customers with the analysis of peptides based on our excellent technology.
The 3D-QSAR method used by Creative Peptides is more physicochemically meaningful and can indirectly reflect the non-bond interaction characteristics between the peptide and the target. Creative Peptides uses a method of comparative molecular similarity indices analysis (CoMSIA) for peptide analysis, the steps of which include:
SAR analysis examines the relationship between a peptide's molecular structure and its biological activity or physicochemical properties. This technique helps optimize peptides for various applications by identifying structural features that enhance their functionality and stability.
SAR analysis enables the identification of key structural features that influence a peptide's performance. By modifying these features, researchers can improve a peptide's stability, specificity, and overall efficacy, which is essential for advancing applications in various industries.
Qualitative SAR identifies structural characteristics that influence activity based on visual observation, while quantitative SAR uses statistical and mathematical models to predict activity. Quantitative SAR provides a more precise, predictive framework for optimizing peptide properties.
QSAR (Quantitative Structure-Activity Relationship) modeling combines molecular descriptors with known bioactivity data to predict the activity or properties of new compounds. This method streamlines the design process, enhancing efficiency and accuracy in peptide optimization.
3D-QSAR provides a deeper understanding of the non-bonded interactions between peptides and their targets, allowing for more informed structural modifications. This method helps visualize complex interactions and guides the design of peptides with improved properties and targeting capabilities.
SAR analysis can optimize peptide materials by identifying structural factors that influence their interactions with surfaces, stability under different conditions, and overall performance. This makes SAR crucial in developing novel peptide-based biomaterials, coatings, and sensors.
SAR analysis helps fine-tune peptide sequences to improve their resistance to environmental factors such as temperature, pH, and enzymatic degradation. This optimization increases the reliability and effectiveness of peptides in industrial applications like biotechnology, agriculture, and material science.
SAR studies enable the design of peptides with enhanced specificity for their targets by adjusting structural elements. These optimizations improve the precision of peptide interactions, making them more effective in applications such as molecular recognition, bioengineering, and environmental science.

References