2018-05-04
Brief information of bombesin
Bombesin is a tetradecapeptide which was originally isolated from Bombina Bombina frog skin then later found to have a wide distribution and range action in mammals. Bombesin belongs to a family of small (10-27 amino acids) peptides which share a highly conserved C-terminal domain and similar biological activities. These analogs differ from bombesin both in chain length at the N-terminus and in selective amino acid substitutions. Litorin and ranatensin are amphibian peptides which contain the C-terminus octapeptide sequence of bombesin with a single substitution. Gastrin-releasing peptide (GRP), neuromedin B (NMB), and neuromedin C (NMC) are mammalian peptides. These peptides can be divided into three subfamilies: the bombesin subfamily which includes GRP and NMC, the ranatensin subfamily which includes NMB, and the phyllolitorin subfamily.
| CAT# | B07014 |
| Product Name | Bombesin |
| CAS No. | 31362-50-2 |
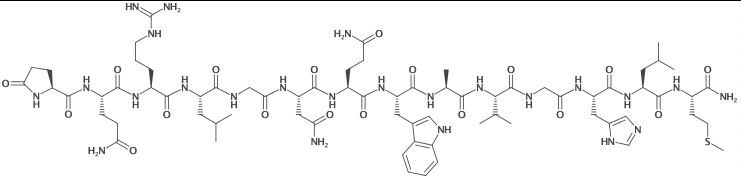
| Sequence | Pyr-QRLGNQWAVGHLM-NH2 |
| M.W/Mr. | 1619.85 |
| Molecular Formula | C71H110N24O18S |
Brief information of bombesin-like peptides
Bombesin-like peptides are initially translated as prepropeptides then processed to yield their biologically active form. For GRP, the signal peptidase cleaves off the “pre” peptide region. Then trypsin-like activity followed by carboxypeptidase B activity cleaves the resulting propeptide at the carboxyl terminus to yield a peptide with a carboxyl-terminal glycine. The glycine is subsequently cleaved by an amidation enzyme which results in the amidated carboxyl terminal methionine and the biologically active form of GRP. The decapeptide NMC results from the cleavage of GRP.
Expression and biological action of bombesin-like peptides
BLPs elicit a wide spectrum of biological responses including cell growth, secretion of gastric acid and a variety of hormones from the stomach, intestine, pancreas, adrenals and pituitary. They also play important roles in regulating circadian rhythms, smooth muscle contraction, body temperature, glucose levels, satiety and certain behaviors such as grooming.
Bombesin-like peptides in mammals are distributed in both the central nervous system (CNS) and the peripheral nervous system. In rat CNS, NMB mRNA is found mostly in the olfactory bulb, dentate gyrus, and the dorsal root ganglion, whereas GRP mRNA is observed mainly in the forebrain. The differential mRNA lacolization suggests that these two neuropeptides have very distinct CNS functions, some of which include thermoregulation, appetite suppression, and behavioral changes. In the peripheral nervous system, bombesin-like peptides are localized in enteric neurons throughout the gastrointestinal tract (GI). Release of these peptides causes smooth muscle cell contraction, GI hormone secretion, and cell proliferation.
Bombesin-like peptides are also expressed in endocrine-like cells in several tisssues including lung, thyroid and pituitary. In the lung and thyroid, bombesin-like peptides are predominantly expressed during the fetal and neonatal developmental stages, respectively, and function as growth factors. In adult tissues, bombesin-like peptides regulate cell growth in lung and hormone secretion in pituitary. Bombesin-like peptides are also localized in reproductive tissues. In the female, these peptides are found in pregnant uterus and have been shown to stimulate uterine contraction and DNA synthesis. In the male, bombesin-like peptides are localized in the prostate gland where they cause contraction and are also secreted in seminal plasma. Several biological responses are elicited by bombesin-like peptides in various model cell lines including cell proliferation in Swiss 3T3 fibroblasts, human bronchial epithelial cells, and human small cell lung cancer (SCLC) cells; prolactin secretion in rat GH4C1 pituitary tumor cells; amylase secretion in rat AR42J pancreatic cells; and insulin secretion in hamster HIT-T15 pancreatic cancer cells.
Reference:
Williams, B. Y. R. H. (1995). Regulation of the gastrin-releasing peptide/bombesin receptor.