2018-04-02
Glucagon is a 29-amino acid peptide hormone that is synthesized in pancreatic α cells from the proglucagon precursor by prohormone convertase 2 (PC2). The primary structure of glucagon is identical in most mammals, with the exception of some amino acid differences in guinea pigs and non-mammalian vertebrates. Glucagon has several physiological functions, including hepatic glucose homeostasis, inotropic and chronotropic activity in the heart, spasmolytic effects on gastrointestinal muscle, lipolytic effects, vasodilatory effects, stimulation of glomerular filtration rate, and regulation of ion transport and electrolyte excretion in the kidney.
Glucose homeostasis
Glucose homeostasis, an important physiological function of glucagon, occurs in hepatocytes through activities that counteract the activities of insulin. In response to low blood sugar, glucagon is released from the pancreas into the bloodstream. Glucagon binds to the glucagon receptor (GR) on hepatocytes, leading to an increase in the hepatocellular content of glucose and the subsequent release of glucose from hepatocytes across the GLUT2 transporter. The effects of glucagon on hepatic glucose synthesis occur at several levels, including regulatory short-term effects on enzymes and long-term effects on transcription. Activation of the GR stimulates signaling pathways involved in glycogenolysis (breakdown of glycogen) and gluconeogenesis (de novo synthesis of glucose from lactate, pyruvate, glycerol, and some amino acids). The effects of glucagon to inhibit glycogen synthesis (glycogenesis) and glucose degradation (glycolysis) are also well established.
Glucagon receptor (GR) signaling pathways in hepatocytes
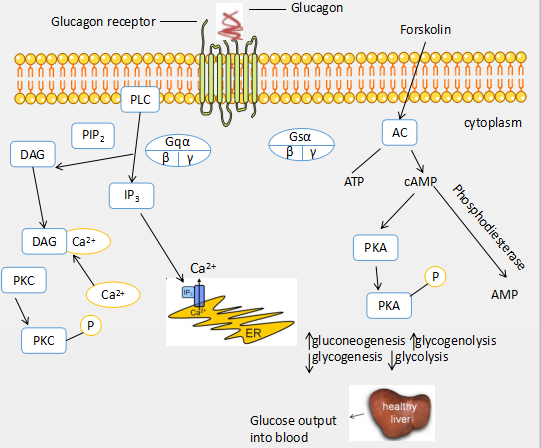
In hepatocytes, glucagon binding to GR activates GPCR signaling pathways involved in glucose metabolism, ultimately leading to increases in blood glucose levels. As for all GPCRs, the binding of glucagon to GR is believed to induce conformational changes in GR that constitute activation of the receptor. The conformational changes may involve reorganization of extracellular loops of GR to accommodate ligand binding, and alterations in interactions of transmembrane helices that affect the cytoplasmic side of GR, allowing intracellular loop regions of the receptor to interact with and activate G proteins.
Upon glucagon binding, the GR stimulates activation of the heterotrimeric G protein Gs, leading to activation of adenylyl cyclase (AC) and a resultant increase in cyclic AMP (cAMP), which activates protein kinase A (PKA) pathways involved in directly increasing glucose production and release from hepatocytes, including glycogenolysis (glycogen degradation to glucose) and gluconeogenesis (de novo glucose synthesis). Additionally, there is a reduction in the use of glucose via a reduction in glycolysis (glucose degradation) and glycogenesis (glycogen synthesis from glucose). Glucagon stimulates glycogenolysis and gluconeogenesis through the regulation of glycogen phosphorylase kinase, glycogen phosphorylase, and 6-phosphofructo-2- kinase/fructose-2,6-bisphosphatase.
Glucagon inhibits glycogenesis and glycolysis through the regulation of glycogen synthase and pyruvate kinase. GR also activates the heterotrimeric G proteins Gq and Gi. Gq activates phospholipase C-beta (PLC-β) to produce inositol 1,4,5-trisphosphate (IP3) and diacylglycerol (DAG) from phosphatidylinositol 4,5-bisphosphate (PIP2). IP3 stimulates the release of calcium from intracellular storage sites, such as the endoplasmic reticulum (ER), which leads to an increase in intracellular calcium and stimulates the activities that increase glucose release from hepatocytes. DAG is involved in stimulation of protein kinase C (PKC). Activated PKC may be directly or indirectly involved in the regulation of the GR, as previously shown. While Gi also appears to be involved in glucagon-mediated intracellular calcium elevation, the exact role of this heterotrimeric G protein in glucagon signaling is not clear. Another downstream target of glucagon signaling is the cAMP-binding protein exchange protein activated by cAMP (Epac). Epac is a recently discovered target of cAMP that is activated independently of PKA, and activates Rap1. Epac signaling affects a multitude of cellular processes, including calcium signaling, cell proliferation, cell survival, cell adhesion, and cell differentiation and has been shown to be a target of glucagon signaling.
Proliferative effects of glucagon in other cell types
While glucagon has generally been associated with a reduction of cell proliferation in hepatocytes, particularly at high concentrations, glucagon stimulates the proliferation of other cell types. Glucagon stimulates the proliferation of cultured human colorectal cancer cells, gastrointestinal cells, and breast cancer cells. As the growth effects of glucagon in these studies were often observed in the presence of serum, it is possible that a growth factor in the serum (e.g., EGF) was interacting with glucagon, as has been reported for the few studies reporting proliferative effects of glucagon/EGF in hepatocytes at low concentrations of glucagon.
Reference:
Nguyen, A. C. (2011). Analysis of the Role of Glucagon in Hepatocellular Carcinoma Cell Growth, Invasion, and Migration and the Regulation of Glucagon Receptor Signaling (Doctoral dissertation, The George Washington University).