2017-11-23
Since the discovery of Substance P (SP) in the early 1930s, its pharmacological actions have been extensively studied. SP has been identified as a neurotransmitter and neuromodulator in the central nervous system (CNS). It has also been shown to be involved in the generation or progression of various physiological and pathophysiological conditions such as pain and depression and a variety of neurodegenerative disorders such as Alzheimer’s, Parkinson’s, Huntington’s, and schizophrenia. Changes in SP concentrations and PPT-A gene expression have been observed in brains of patients suffering from these neurodegenerative diseases. SP has been shown to improve memory and counter age-related performance deficits in animals. It works in concert with several hormones and other endogenous substances such as lutenizing hormone, angiotensin II, and serotonin, thus modulating their actions. Many of these effects require the selective passage of SP across the blood-brain barrier (BBB) following its release in the periphery.
| CAT# | 10-101-52 |
| Product Name | Substance P Acetate |
| CAS No. | 33507-63-0 |
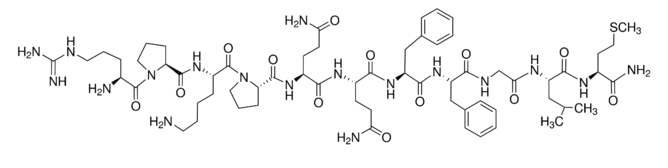
| Sequence | H-Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-NH2 |
| M.W/Mr. | 1347.63 |
| Molecular Formula | C63H98N18O13S |
Substance P (SP), neurokinin A (NKA), and neurokinin B (NKB) belong to a family of mammalian tachykinin peptides characterized by the presence of a common C-terminal amidated sequence (Phe-X-Gly-Leu-Met-NH2) in their structure which is responsible for their fundamental interaction with their corresponding receptors, neurokinin-1 (NK-1), neurokinin-2 (NK-2), and neurokinin-3 (NK-3), respectively. cDNA and genomic cloning experiments indicate that SP and NKA are derived from differential RNA splicing of the preprotachykinin-A (PPT-A) gene. NKB is encoded by the preprotachykinin-B (PPT-B) gene. Although these three neuropeptides and their corresponding receptors have extensive cross-talk amongst each other, SP and the NK-1 receptor is considered the prototype for the interactions between the tachykinin peptide family and its receptors in the central nervous system.
Methamphetamine and Substance P
Aside from their favorable anatomical positions in the basal ganglia, SP and NK-1 receptor have been shown to have functional modulatory effects on the dopaminergic and glutamatergic systems. Thus, indicating that the pair may serve as key participants in the deleterious effects of methamphetamine (METH). METH treatment has been shown to elevate the striatal and nigral concentrations of SP neuropeptide and increase PPT-A mRNA within striatonigral neurons. These increases follow the elevation of extracellular DA levels. Microinjections of SP in the SN and microperfusions of a NK-1 receptor agonist in the striatum results in the increased release of DA and glutamate in the striatum. Also, the firing of SP-containing neurons in the striatum can increase the firing probability of cholinergic and SST/NOS interneurons by receptor-mediated opening of tetrodotoxin-insensitive ion channels. Local perfusion of SP in the striatum can elevate extracellular acetylcholine through NK-1 receptor mediated mechanisms. Blockade of the NK-1 receptor prior to METH treatment prevents METH-induced loss of dopamine transporters, DA content, TH, as well as the strong induction of reactive gliosis. Moreover, there is evidence of interactions between glutamate and stimulation of the NK-1 receptor, with subsequent protein kinase C–dependent facilitation of NMDA transmission as well as the formation of NO. Intrastriatal infusion of NMDA receptor antagonist, CPP, decreased SP mRNA expression in the striatum in a dose-dependent manner. Pharmacological blockade of the NK-1 receptor and genetic knockouts of the substance P gene, preprotachykinin-A (PPT-A), showed reduction and abrogation of excitotoxin-induced seizures. Also, mice lacking the PPT-A do not display kainate-induced cell death in the hippocampus. In addition, activation the NK-1 receptor by SP on SST/NOS-containing interneurons may lead to the activation of NOS to synthesize nitric oxide (NO). NO synthesized in the interneurons, can readily diffuse through the membrane and enter the spines of the striatal projection neurons inducing the synthesis of cGMP and indirectly potentiate the release and actions of glutamate.
Reference:
Zhu, J. P. Q. (2005). Methamphetamine-induced cell death in the striatum and the role of neurokinin-1 receptor in its pathogeny.