GMP-compatible Peptide Standards13C 15N Labeled PeptidesMethod ValidationBiomarker Quantification
At Creative Peptides, we specialize in the custom synthesis of stable isotope labeled peptides, providing high-precision internal standards and reference materials for quantitative proteomics, bioanalysis, and regulated pharmaceutical research. By incorporating non-radioactive isotopes such as 13C, 15N, and 2H at defined amino acid positions, our expert chemists deliver peptides with identical chemical behavior to native counterparts while enabling accurate mass differentiation by LC-MS/MS. Serving pharmaceutical companies, biotechnology enterprises, and contract research organizations worldwide, we offer scalable, reproducible, and fully documented isotope-labeled peptide solutions to support method development, validation, and routine analysis.
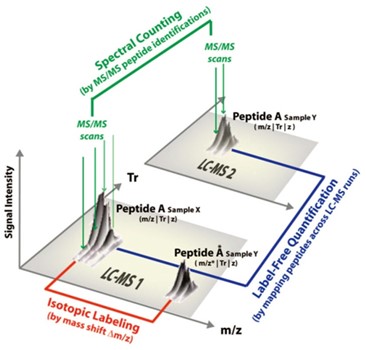 Overview of quantification approaches in LC-MS based proteomic experiments.
Overview of quantification approaches in LC-MS based proteomic experiments.Quantitative protein and peptide analysis in complex biological samples is often limited by matrix effects, ion suppression, variable digestion efficiency, and instrument-related signal fluctuation.
Stable isotope labeled peptides directly address these analytical challenges by:
We offer enterprise-focused stable isotope labeled peptide solutions designed to support quantitative proteomics, regulated bioanalysis, and long-term biomarker programs. Our services are built around the real-world needs of pharmaceutical companies, biotechnology enterprises, and CROs—where analytical accuracy, reproducibility, documentation, and supply continuity are critical. Each project is supported by experienced peptide chemists and bioanalytical specialists with deep understanding of LC-MS/MS–based quantification workflows.
Selecting the right peptide standard is essential for reliable quantitative analysis. Our team supports customers at the assay-design stage to ensure isotope-labeled peptides are fit for purpose.
This consultative approach reduces assay redevelopment risk and accelerates method validation timelines.
We manufacture stable isotope labeled peptides using solid-phase peptide synthesis with qualified isotope-labeled amino acid building blocks to ensure accurate incorporation and consistent performance.
All products are suitable for use as internal standards in complex biological matrices.
To accurately reflect endogenous analytes, we supply isotope-labeled peptides incorporating biologically or analytically relevant modifications.
These solutions are commonly applied in translational research, biomarker qualification, and preclinical assay development.
Each isotope-labeled peptide is analytically qualified to ensure suitability for quantitative and regulated workflows.
Documentation is designed to support internal QA review, method validation, and regulatory submissions.
For enterprise programs requiring long-term consistency, we provide structured supply and lifecycle management solutions.
Selecting an appropriate isotope labeling strategy is a foundational decision in quantitative LC-MS/MS assay design. The choice influences mass separation, chromatographic behavior, method robustness, and long-term assay transferability. Enterprise laboratories typically evaluate labeling strategies based on analytical risk, matrix complexity, regulatory expectations, and program scale.
| Labeling Strategy | Common Isotopes Used | Typical Use Case | Why Enterprise Customers Choose It | Key Considerations |
|---|---|---|---|---|
| Single-residue heavy label | 13C / 15N Lys or Arg | Targeted SRM/MRM or PRM assays | Predictable mass shift and co-elution for routine quantification | Confirm sufficient separation from natural isotopic envelope |
| Multi-residue heavy label | 13C / 15N on multiple residues | Complex matrices or interference-prone assays | Improves selectivity and signal discrimination | Balance increased cost vs. analytical benefit |
| Uniform peptide labeling | Multiple 13C / 15N residues | Absolute quantification in highly complex samples | Maximum mass separation and inter-lab robustness | May be unnecessary for high-resolution instruments |
| Deuterium labeling | 2H (D) | Cost-sensitive or legacy workflows | Lower material cost in some applications | Potential retention time shift due to isotope effects |
| Hybrid labeling | 13C / 15N ± 2H | Custom or regulated biomarker assays | Flexible mass engineering for critical targets | Requires careful isotopic pattern validation |
Stable isotope labeled peptide standards are deployed in different formats depending on assay design, analytical complexity, and regulatory context. Enterprise users typically select standard types based on how well they correct for variability across digestion, sample preparation, and LC-MS analysis.
| Standard Type | Where It's Used | What It Controls For | Typical Enterprise Use Case | Method Development Notes |
|---|---|---|---|---|
| AQUA-style heavy peptide | Targeted LC-MS/MS quantification | Ionization and matrix effects | Biomarker validation, translational research | Sequence and co-elution must match endogenous peptide |
| Multiplex peptide panels | Multi-analyte MRM/PRM assays | Run-to-run and inter-analyte variability | CRO studies, pathway profiling | Standardize concentration ranges across analytes |
| PTM-matched standards | PTM-specific quantification | Modification-dependent behavior | Signaling and MOA studies | Exact PTM site and chemistry must be matched |
| Surrogate peptides | Early discovery screens | Partial process variability | Feasibility and pilot assays | Not suitable for regulated quantification |
| Extended or domain peptides | Enrichment-based workflows | Recovery and processing bias | Immunoaffinity or pull-down assays | Confirm compatibility with enrichment reagents |
Post-Translational Modification often occur at low stoichiometry and are sensitive to sample handling and enrichment bias. Stable isotope labeled PTM peptides provide modification-specific internal standards that enable accurate, site-resolved quantification in both research and regulated analytical settings.
| PTM Type | Biological Context | Analytical Challenge | Benefit of Heavy Standard | Practical Notes |
|---|---|---|---|---|
| Phosphorylation | Cell signaling, kinase activity | Low abundance, enrichment variability | Enables site-specific, reproducible quantification | Protect against dephosphorylation during handling |
| Oxidation | Stress response, sample artifacts | Mixed oxidized/unoxidized populations | Supports accurate ratioing of oxidized forms | Define oxidation state explicitly |
| Acetylation | Epigenetics, protein regulation | Isobaric interference | Improves confidence in site localization | Confirm fragmentation strategy |
| Methylation | Chromatin and transcription control | Multiple methyl states | State-specific quantification | Separate mono/di/tri forms chromatographically |
| Deamidation | Stability and aging studies | Process-induced conversion | Controls analytical artifacts | Document handling conditions carefully |
Robust quality control and transparent documentation are essential for enterprise and regulated applications. Stable isotope labeled peptides used as quantitative standards must be fully characterized to ensure identity, purity, labeling integrity, and long-term reproducibility across studies and sites.
| QC / Documentation Item | Typical Method | What It Confirms | Enterprise Value | Deliverable |
|---|---|---|---|---|
| Identity confirmation | LC-MS | Correct sequence and molecular weight | Prevents assay failure due to wrong analyte | MS summary in CoA |
| Isotopic labeling verification | Isotopic pattern analysis | Correct isotope incorporation | Ensures distinguishable internal standard | Labeling statement |
| Purity assessment | Analytical HPLC | Impurity profile | Reduces quantification bias | HPLC chromatogram |
| Content assignment | UV or amino acid analysis | Peptide amount consistency | Critical for absolute quantification | Content statement |
| Co-elution check | LC comparison | Comparable chromatographic behavior | Valid internal standard correction | RT confirmation note |
| Traceability | Batch documentation | Lot and manufacturing history | Supports audits and long-term programs | Certificate of Analysis |
| Storage & handling guidance | Stability-informed recommendations | Integrity over time | Prevents revalidation and material loss | Handling instructions |
Quantification-Focused Design
Peptides are designed specifically for quantitative LC-MS/MS workflows, ensuring co-elution and reliable internal standard performance.
High Isotopic Integrity
Use of qualified isotope-labeled amino acids delivers consistent mass shifts and reproducible isotopic distributions.
Application-Driven Expertise
Deep experience supporting biomarker validation, proteomics panels, and regulated bioanalytical assays.
Batch-to-Batch Consistency
Manufacturing controls and documentation support longitudinal studies and inter-site comparability.
Comprehensive QC & Documentation
Each peptide is supplied with a detailed Certificate of Analysis suitable for enterprise and regulated workflows.
Scalable Supply Capability
From milligram research quantities to program-level supply, supporting method validation and routine analysis.
Enterprise-Oriented Support
Familiarity with QA review, procurement requirements, and cross-functional program coordination.
Long-Term Program Reliability
Structured lifecycle and repeat-supply support reduces revalidation risk and operational disruption.
Trusted Partner Approach
Acting as a technical partner rather than a commodity supplier for critical quantitative standards.
Our workflow for stable isotope labeled peptide projects is designed to ensure analytical accuracy, traceability, and consistency across discovery, development, and regulated bioanalytical stages. Each step is executed with close alignment to the customer's assay design, validation strategy, and long-term supply requirements.
1
Project Consultation & Assay Alignment
2
Peptide Design & Isotope Labeling Strategy Confirmation
3
Stable Isotope Labeled Peptide Synthesis
4
Analytical Characterization & Quality Verification
5
Delivery, Documentation & Lifecycle Support
Stable isotope labeled peptides are widely used as internal standards and reference materials to enable accurate, reproducible quantification by LC-MS/MS. They support enterprise workflows across quantitative proteomics, bioanalysis, biomarker validation, and regulated method development by correcting for variability introduced during sample preparation, separation, and mass spectrometric detection.
 Stable isotope labeled peptides are used to enhance LC-MS quantification accuracy by comparing light and heavy peptide standards, enabling precise concentration measurements. This process includes sample preparation, LC separation, mass spectrometry detection, and data analysis to obtain accurate quantitative results.
Stable isotope labeled peptides are used to enhance LC-MS quantification accuracy by comparing light and heavy peptide standards, enabling precise concentration measurements. This process includes sample preparation, LC separation, mass spectrometry detection, and data analysis to obtain accurate quantitative results.
If your team needs reliable stable isotope labeled peptide standards for quantitative proteomics, biomarker verification, PTM analysis, or regulated LC-MS/MS method development, Creative Peptides can support your program with application-aligned design, controlled manufacturing, and thorough analytical qualification. Contact us to discuss peptide sequences, labeling strategy, purity requirements, documentation expectations, and long-term supply planning. Contact us today to request a technical consultation and quotation.
Stable isotope-labeled peptides act as internal standards, enabling accurate quantification of target peptides or proteins. These labeled peptides improve detection sensitivity and dynamic range in mass spectrometry, while also allowing for multiplexing by using different isotopic labels to distinguish multiple samples within a single experiment, facilitating high-throughput analysis.
Stable isotope-labeled peptides are typically synthesized using solid-phase peptide synthesis (SPPS) techniques. During synthesis, amino acids containing stable isotopes are incorporated at specific positions within the peptide sequence. The resulting labeled peptides are then purified and characterized for use in mass spectrometry experiments.
Protein quantification can be approached in different ways, including relative quantification, which compares protein expression levels between different samples or conditions; absolute quantification, which determines the exact concentration of specific proteins; and targeted proteomics, which focuses on quantifying specific peptides or proteins of interest within complex biological samples.
Yes, many providers offer customization services where stable isotope-labeled peptides can be synthesized and optimized according to your specific experimental requirements, including peptide sequence, labeling scheme, and purity.
Turnaround times and costs can vary depending on the complexity of the synthesis, the quantity and purity of labeled peptides required, and the customization options. It's best to inquire with the service provider for specific details regarding turnaround times and pricing.
Yes, stable isotope-labeled peptides can be used with various mass spectrometry platforms, including triple quadrupole, quadrupole-time-of-flight (Q-TOF), and Orbitrap instruments, among others. Compatibility may vary depending on the labeling scheme and specific experimental protocols.

References