2017-04-27
PT-141, also called as Bremelanotide, is a derivative for Melanotan 2 (M2). Unlike M2, PT-141 lacks C-terminal amide molecules. PT-141, a synthetic cyclic hepta-peptide lactam derivative of alpha-Melanocyte-stimulating hormone (alpha-MSH), is an agonist at melanocortin receptors including the MC3R and MC4R, which are expressed primarily in the central nervous system. Melanocortin receptors are members of the rhodopsin family of 7-transmembrane G protein-coupled receptors. There are five known members of the melanocortin receptor system each with differing specificities for melanocortins: MC1R; MC2R; MC3R; MC4R; MC5R.
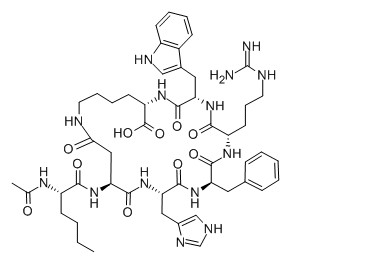
PT-141 is a synthetic peptide with the amino acid sequence Ac-Nle-c[Asp-His-D-Phe-Arg-Trp]Cys-NH2. The structure includes an N-terminal acetylated N-leucine (Ac-Nle) that stabilizes the peptide, a cyclic core composed of aspartic acid (Asp), histidine (His), D-phenylalanine (D-Phe), arginine (Arg), and tryptophan (Trp) arranged in a cyclic sequence, and a C-terminal cysteine with an amidated end (Cys-NH2) that contributes to stability and activity. This cyclic configuration enhances PT-141's interaction with melanocortin receptors, which is crucial for its effects on sexual function.
PT-141 was developed from the peptide hormone melanotan II which underwent testing as a sunless tanning agent. Sunless tanning, also known as UV-free tanning, self tanning, spray tanning (when applied topically), or fake tanning, refers to the application of chemicals to the skin to produce an effect similar in appearance to a suntan. In initial testing, melanotan II did induce the production of darkening dermal pigmentation (melanogenesis) but additionally caused sexual arousal and spontaneous erections as unexpected side effects in nine out of the ten original male volunteer test subjects. Due to this potential, PT-141 is being studied as a potential treatment for sexual disorders such as erectile disorders in men and sexual arousal disorders in women, especially Hypoactive sexual desire disorder (HSDD) and female sexual interest/arousal disorder (FSIAD). However, reports of elevated blood pressures halted the trial.
Hypoactive sexual desire disorder (HSDD) or inhibited sexual desire (ISD) is considered a sexual dysfunction and is characterized as a lack or absence of sexual fantasies and desire for sexual activity, as judged by a clinician. Female sexual arousal disorder (FSAD) is a disorder characterized by a persistent or recurrent inability to attain sexual arousal or to maintain arousal until the completion of a sexual activity. PT-141 is the only synthetic aphrodisiac. Unlike Viagra and other related medications (PDE5 inhibitors), PT-141 does not act upon the vascular system, but directly through the nervous system (hypothalamus) to increase desire. Viagra, Cials and Levitra are not considered aphrodisiacs as they do not have any direct effect on the libido. However, treatment with PDE5 inhibitors and PT-141 are known to have a synergistic effect.
PT-141 offers several benefits, particularly in the realm of sexual health and potential therapeutic applications:
Improvement in Sexual Desire: PT-141 has shown efficacy in increasing sexual desire and arousal, particularly in individuals with hypoactive sexual desire disorder (HSDD), making it a valuable treatment option for those struggling with sexual dysfunction.
Alternative to Traditional ED Treatments: For patients with erectile dysfunction who do not respond well to traditional treatments like Viagra or Cialis, PT-141 offers an alternative approach by working through different mechanisms to enhance sexual arousal.
Quick Onset of Action: PT-141 can provide rapid onset of effects, with some users experiencing benefits within a short period of administration, which is advantageous for both planned and spontaneous sexual activity.
Minimal Systemic Side Effects: Compared to other treatments for sexual dysfunction, PT-141 tends to have fewer systemic side effects, as its action is primarily localized to sexual arousal pathways.
Potential for Broader Applications: Beyond sexual health, PT-141's impact on appetite and the brain's reward systems suggests potential for future research into other therapeutic areas, such as appetite regulation and psychological disorders.
Synthetic Peptide Hormone at Creative Peptides
| Name | CAS | CAT# | Price |
|---|---|---|---|
| Gonadorelin | 33515-09-2 | 10-101-166 | Inquiry |
| GHRP-6 | 87616-84-0 | 10-101-208 | Inquiry |
| Octreotide | 83150-76-9 | R1574 | Inquiry |
| Lanreotide | 108736-35-2 | R812 | Inquiry |
| Exenatide | 141732-76-5 | 10-101-16 | Inquiry |
| Liraglutide | 204656-20-2 | 10-101-59 | Inquiry |
| Oxytocin | O06003 | Inquiry |
In a phase 2B trial, PT-141 was reformulated as a lower dose, subcutaneous injection to measure efficacy for HSDD and/or FSAD treatment in premenopausal women. In a 4-week at-home trial, 1.75 mg PT-141 showed statistically significant improvements as compared with placebo in five measures of FSD: number of satisfying sexual events per month, total and sexual domain scores on the FSFI, and total and desire domain scores on the FSDS. PT-141 was associated with minimal and transient increases in blood pressure (~3 mmHg) that were limited to the first four hours after administration. Protocol-defined blood pressure withdrawal criteria were not met at higher frequency in PT-141-treated subjects than in those taking placebo. Further studies on PT-141 dosing have suggested optimal increases in arousal, desire, and satisfaction with sexual events with 1.25 and 1.75 mg subcutaneous injections. At these doses, adverse events included nausea (22 % and 24 %, respectively), placebo 3 %; flushing (14 % and 17 %, respectively), placebo 0 %; and headache (9 % and 14 %, respectively), placebo 3 %. MC3R and MCR4 receptors are involved in many physiological systems and there may be theoretical risks of activating these receptors. The long-term effects of BMT are unknown.
Bremelanotide works by activating melanocortin receptors in the brain, specifically the melanocortin-4 receptor (MC4R). It is a synthetic peptide developed to treat sexual dysfunction, particularly in premenopausal women with hypoactive sexual desire disorder (HSDD). Here's a more detailed breakdown of its mechanism:
Activation of Melanocortin Receptors: Bremelanotide binds to and activates melanocortin receptors in the brain. The primary receptor involved is the melanocortin-4 receptor (MC4R), which plays a significant role in modulating sexual behavior and desire.
Neurotransmitter Modulation: By stimulating these receptors, Bremelanotide influences the release of neurotransmitters that are involved in sexual arousal and desire. This includes dopamine, a neurotransmitter that is closely associated with pleasure and reward pathways in the brain.
Increased Sexual Arousal: The activation of MC4R and subsequent neurotransmitter release leads to increased sexual arousal and desire. This process is not directly linked to the vascular system, which differentiates Bremelanotide from other sexual dysfunction treatments like sildenafil (Viagra) that primarily work by increasing blood flow to the genital area.
Administration and Absorption: Bremelanotide is typically administered via subcutaneous injection. After injection, it is rapidly absorbed into the bloodstream and crosses the blood-brain barrier to exert its effects on the central nervous system.
Non-Hormonal Approach: Unlike some other treatments for sexual dysfunction that may involve hormonal pathways, Bremelanotide's mechanism is non-hormonal. This makes it a unique option, particularly for women who might not benefit from or cannot use hormone-based therapies.
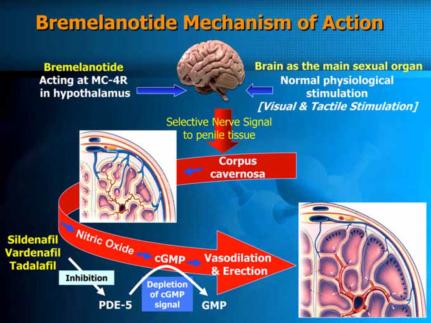 Mechanism of Bremelanotide (Shadiack A M, et al., 2007)
Mechanism of Bremelanotide (Shadiack A M, et al., 2007)
PT-141 generally has effects that last between 6 to 12 hours after administration. The duration and onset of action can vary based on several factors:
Onset of Action: The effects of PT-141 typically begin within 30 minutes to an hour after subcutaneous injection. This rapid onset is beneficial for those seeking prompt results.
Peak Effect: The peak effect of Bremelanotide usually occurs around 1 to 2 hours post-injection. During this time, users may experience the most significant increase in sexual desire and arousal.
Duration of Effect: The duration of the drug's effects can last from 6 to 12 hours. This extended period allows for flexibility in timing and can enhance sexual spontaneity.
Individual Variability: The duration can vary significantly between individuals due to factors such as metabolism, overall health, body weight, and individual response to the drug. Some users may find the effects last longer or shorter than the average range.
Dosage: The standard dosage of Bremelanotide is typically 1.75 mg administered via subcutaneous injection. Variations in dosage can influence both the onset and duration of effects. Higher doses might prolong the duration, while lower doses could shorten it.
Frequency of Use: The frequency of PT-141 administration can also impact its efficacy and duration. Regular use might lead to a more predictable response, whereas occasional use might result in variable duration and onset.
Absorption and Distribution: After subcutaneous injection, Bremelanotide is rapidly absorbed into the bloodstream and distributed throughout the body. The pharmacokinetics of the drug, including its half-life (approximately 2.7 hours), play a role in determining how long its effects last.
Metabolic Factors: Individual metabolic rate and liver function can affect how quickly the drug is broken down and eliminated from the body, influencing the overall duration of its effects.
Tolerance and Sensitivity: Over time, some users may develop a tolerance to PT-141, potentially requiring higher doses to achieve the same effect. Conversely, individuals with heightened sensitivity to the drug may experience longer-lasting effects even at standard doses.
Here are some key applications of PT-141:
Sexual Dysfunction Treatment: PT-141 is approved by the FDA under the brand name Vyleesi for the treatment of premenopausal women with hypoactive sexual desire disorder (HSDD). It works by activating melanocortin receptors, which can enhance sexual desire and arousal. Although not FDA-approved for men, PT-141 has been studied for its potential to treat erectile dysfunction (ED) in men, particularly in cases where traditional treatments are ineffective. It has shown promise in clinical trials for improving sexual arousal and satisfaction.
Appetite Regulation: PT-141 affects the brain's reward pathways and may influence appetite. While its primary use is not weight loss, its effect on appetite regulation has led to some exploration of its potential in weight management strategies.
Neurobiology Studies: PT-141 is used in research to better understand the mechanisms of sexual arousal and desire. By studying its effects, scientists can gain insights into the neurobiological pathways involved in sexual function and dysfunction.
Psychological Disorders: Preliminary research suggests that PT-141 might have potential in treating psychological conditions related to sexual arousal and desire. However, these applications are still in the exploratory stages and require further research.
References