Peptide purification refers to the process of isolating and purifying peptides from a mixture. Peptides are short chains of amino acids linked together by peptide bonds. They are synthesized by methods such as solid phase peptide synthesis or recombinant DNA technology. However, these synthesis processes usually result in peptide mixtures containing impurities, by-products, protecting groups and reagents used in the synthesis reaction. Peptide purification is essential to obtain high purity peptides for further characterization, biological research or therapeutic applications. It involves isolating the desired peptide from impurities and obtaining high yield and quality purified peptide samples.
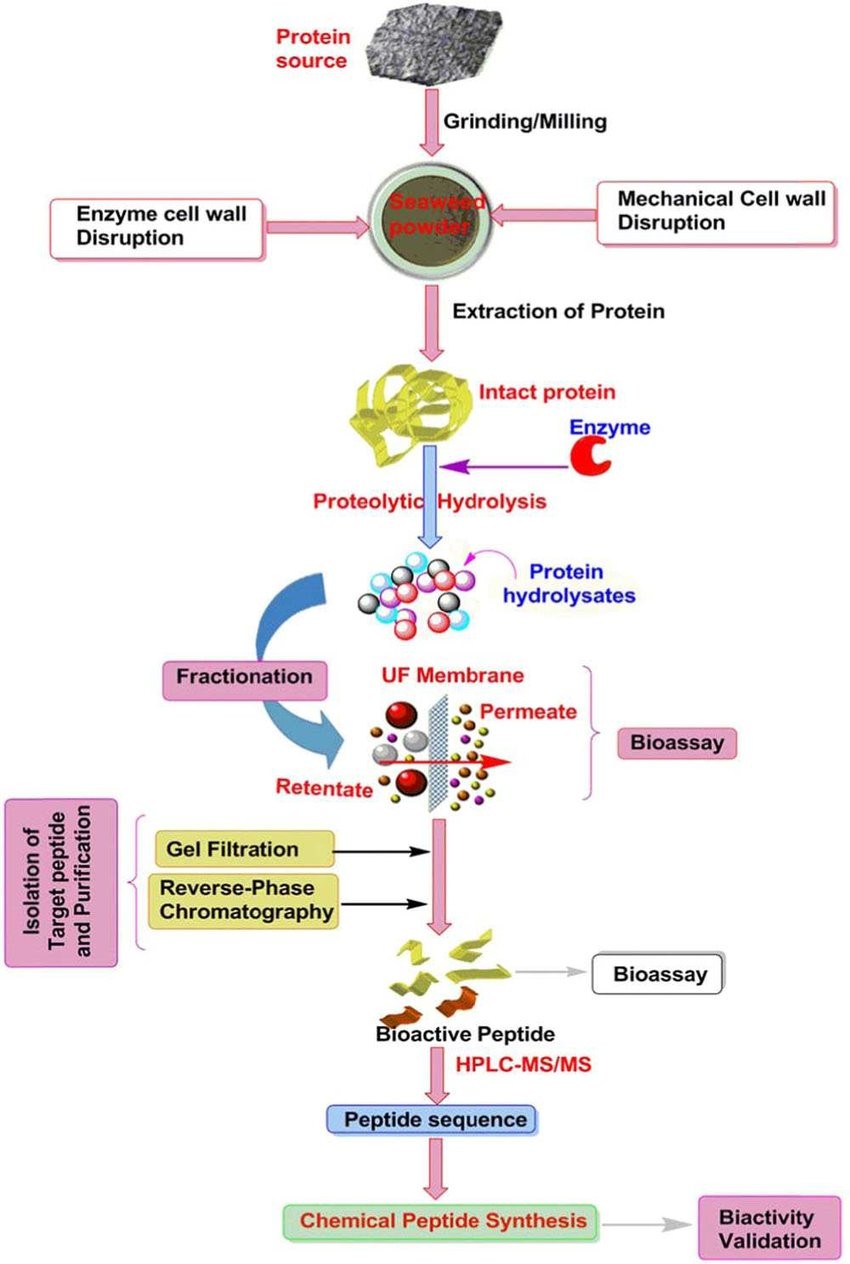 Fig. 1. Extraction, purification and isolation procedures of bioactive peptides (Journal of Food Science. 2018, 83(1): 6-16).
Fig. 1. Extraction, purification and isolation procedures of bioactive peptides (Journal of Food Science. 2018, 83(1): 6-16).
The first critical step is cleavage and deprotection, where the peptide is released from the resin and side-chain protecting groups are removed using a strong acid cocktail like TFA.
Purification removes synthesis by-products (deletion sequences, truncated peptides), reagents, and impurities to achieve the high purity required for reliable research and consistent biological activity.
Preparative Reversed-Phase High-Performance Liquid Chromatography (RP-HPLC) is the industry standard, separating peptides based on their hydrophobicity with high resolution.
Conditions are optimized using analytical HPLC runs to find the mobile phase (e.g., water/acetonitrile with TFA) and gradient that best separate the target peptide from its impurities.
Hydrophobic peptides may have poor solubility in aqueous buffers, requiring alternative solvents like DMSO or isopropanol in the mobile phase, or the use of ion-pairing agents.
MS analysis before, during, and after purification confirms the identity of the target peptide and monitors for the removal of impurities with similar molecular weights.

Reference